Host-Parasite Interactions in Toxoplasma gondii-Infected Cells: Roles of Mitochondria, Microtubules, and the Parasitophorous Vacuole
- PMID: 39769222
- PMCID: PMC11677533
- DOI: 10.3390/ijms252413459
Host-Parasite Interactions in Toxoplasma gondii-Infected Cells: Roles of Mitochondria, Microtubules, and the Parasitophorous Vacuole
Abstract
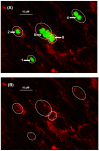
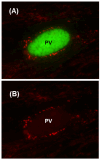
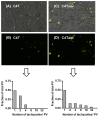
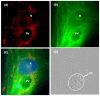
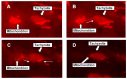
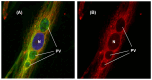
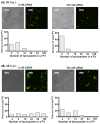
An intracellular protozoan, the Apicomplexan parasite Toxoplasma gondii (T. gondii) infects nucleated cells, in which it triggers the formation of a specialized membrane-confined cytoplasmic vacuole, named the parasitophorous vacuole (PV). One of the most prominent events in the parasite's intracellular life is the congregation of the host cell mitochondria around the PV. However, the significance of this event has remained largely unsolved since the parasite itself possesses a functional mitochondrion, which is essential for its replication. Here, we explore several fundamental aspects of the interaction between the PV and the host cell mitochondria. They include the detailed features of the congregation, the nature and mechanism of the mitochondrial travel to the PV, and the potential significance of the migration and congregation. Using a combination of biochemical assays, high-resolution imaging, and RNAi-mediated knockdown, we show that: (i) mitochondrial travel to the PV starts very early in parasite infection, as soon as the smallest PV takes shape; (ii) the travel utilizes the contractile microtubular network of the host cell; and (iii) near the end of the parasitic life cycle, when most PVs have reached their largest sustainable size and are about to lyse in order to release the progeny parasites, the associated mitochondria change their usual elongated shape to small spheres, apparently resulting from increased fission. Intriguingly, despite the well-known mitochondrial role as a major producer of cellular ATP, the parasite does not seem to use cellular mitochondrial ATP. Together, these findings may serve as foundations for future research in host-parasite interaction, particularly in the elucidation of its mechanisms, and the possible development of novel antiparasitic drug regimens.
Keywords: Apicomplexa; Toxoplasma; energy metabolism; microtubule; mitochondria; protozoa.
Conflict of interest statement
This work formed part of a thesis of Joel Andrews, submitted in 2010 to the Graduate Faculty of the University of South Alabama in partial fulfillment of Doctor of Philosophy (Ph.D.) in Basic Medical Sciences (Mentor: Sailen Barik, Chair of the Dissertation Committee). The authors declare no conflicts of interest.
Figures










Similar articles
-
Biogenesis of and activities at the Toxoplasma gondii parasitophorous vacuole membrane.Subcell Biochem. 2008;47:155-64. doi: 10.1007/978-0-387-78267-6_12. Subcell Biochem. 2008. PMID: 18512349 Review.
-
Toxoplasma gondii salvages sphingolipids from the host Golgi through the rerouting of selected Rab vesicles to the parasitophorous vacuole.Mol Biol Cell. 2013 Jun;24(12):1974-95. doi: 10.1091/mbc.E12-11-0827. Epub 2013 Apr 24. Mol Biol Cell. 2013. PMID: 23615442 Free PMC article.
-
Toxoplasma gondii association with host mitochondria requires key mitochondrial protein import machinery.Proc Natl Acad Sci U S A. 2021 Mar 23;118(12):e2013336118. doi: 10.1073/pnas.2013336118. Proc Natl Acad Sci U S A. 2021. PMID: 33723040 Free PMC article.
-
[Formation and diversity of parasitophorous vacuoles in parasitic protozoa. The Coccidia (Sporozoa, Apicomplexa)].Tsitologiia. 2003;45(4):339-56. Tsitologiia. 2003. PMID: 14520865 Review. Russian.
-
GRA proteins of Toxoplasma gondii: maintenance of host-parasite interactions across the parasitophorous vacuolar membrane.Korean J Parasitol. 2009 Oct;47 Suppl(Suppl):S29-37. doi: 10.3347/kjp.2009.47.S.S29. Korean J Parasitol. 2009. PMID: 19885333 Free PMC article. Review.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

