Boosting Lipofection Efficiency Through Enhanced Membrane Fusion Mechanisms
- PMID: 39769303
- PMCID: PMC11677079
- DOI: 10.3390/ijms252413540
Boosting Lipofection Efficiency Through Enhanced Membrane Fusion Mechanisms
Abstract
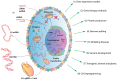
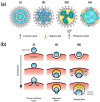
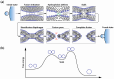
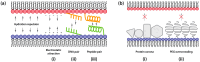
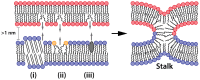
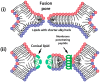
Gene transfection is a fundamental technique in the fields of biological research and therapeutic innovation. Due to their biocompatibility and membrane-mimetic properties, lipid vectors serve as essential tools in transfection. The successful delivery of genetic material into the cytoplasm is contingent upon the fusion of the vector and cellular membranes, which enables hydrophilic polynucleic acids to traverse the hydrophobic barriers of two intervening membranes. This review examines the critical role of membrane fusion in lipofection efficiency, with a particular focus on the molecular mechanisms that govern lipoplex-membrane interactions. This analysis will examine the key challenges inherent to the fusion process, from achieving initial membrane proximity to facilitating final content release through membrane remodeling. In contrast to viral vectors, which utilize specialized fusion proteins, lipid vectors necessitate a strategic formulation and environmental optimization to enhance their fusogenicity. This review discusses recent advances in vector design and fusion-promoting strategies, emphasizing their potential to improve gene delivery yield. It highlights the importance of understanding lipoplex-membrane fusion mechanisms for developing next-generation delivery systems and emphasizes the need for continued fundamental research to advance lipid-mediated transfection technology.
Keywords: cationic lipids; fusion pore; lipid-based vector; membrane fusion; nanotechnology; transfection.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Lipoplex morphologies and their influences on transfection efficiency in gene delivery.J Control Release. 2007 Nov 20;123(3):184-94. doi: 10.1016/j.jconrel.2007.08.022. Epub 2007 Aug 24. J Control Release. 2007. PMID: 17913276 Review.
-
Single histidine residue in head-group region is sufficient to impart remarkable gene transfection properties to cationic lipids: evidence for histidine-mediated membrane fusion at acidic pH.Gene Ther. 2003 Aug;10(15):1206-15. doi: 10.1038/sj.gt.3301979. Gene Ther. 2003. PMID: 12858185
-
Improved in vitro gene transfer mediated by fluorinated lipoplexes in the presence of a bile salt surfactant.J Gene Med. 2001 Jul-Aug;3(4):338-44. doi: 10.1002/jgm.188. J Gene Med. 2001. PMID: 11529663
-
Nature as a source of inspiration for cationic lipid synthesis.Genetica. 2010 Feb;138(2):153-68. doi: 10.1007/s10709-009-9405-8. Epub 2009 Sep 11. Genetica. 2010. PMID: 19763845 Review.
-
A molecular view on the escape of lipoplexed DNA from the endosome.Elife. 2020 Apr 16;9:e52012. doi: 10.7554/eLife.52012. Elife. 2020. PMID: 32297853 Free PMC article.
Cited by
-
Transfection Technologies for Next-Generation Therapies.J Clin Med. 2025 Aug 5;14(15):5515. doi: 10.3390/jcm14155515. J Clin Med. 2025. PMID: 40807133 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

