Nanosilver-Biopolymer-Silica Composites: Preparation, and Structural and Adsorption Analysis with Evaluation of Antimicrobial Properties
- PMID: 39769310
- PMCID: PMC11679571
- DOI: 10.3390/ijms252413548
Nanosilver-Biopolymer-Silica Composites: Preparation, and Structural and Adsorption Analysis with Evaluation of Antimicrobial Properties
Abstract
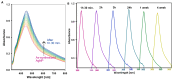
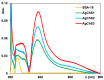
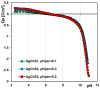
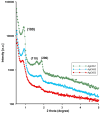
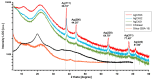
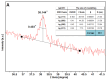
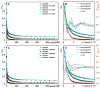
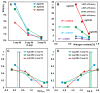
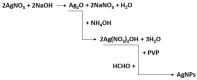
In this article, we report on the research on the synthesis of composites based on a porous, highly ordered silica material modified by a metallic nanophase and chitosan biofilm. Due to the ordered pore system of the SBA-15 silica, this material proved to be a good carrier for both the biologically active nanophase (highly dispersed silver nanoparticles, AgNPs) and the adsorption active phase (chitosan). The antimicrobial susceptibility was determined against Gram-positive Staphylococcus aureus ATCC 25923, Gram-negative bacterial strains (Escherichia coli ATCC 25922, Klebsiella pneumoniae ATCC 700603, and Pseudomonas aeruginosa ATCC 27853), and yeast Candida albicans ATCC 90028. The zones of microbial growth inhibition correlated with the content of silver nanoparticles deposited in the composites and were the largest for C. albicans (14-21 mm) and S. aureus (12-17 mm). The suitability of the composites for the purification of water and wastewater from anionic pollutants was evaluated based on kinetic and equilibrium adsorption studies for the dye Acid Red 88. The composite with the highest amount of the chitosan component showed the greatest adsorption capacity (am) of 0.57 mmol/g and the most effective kinetics with a rate constant (log k) and half-time (t0.5) of -0.21 and 1.62 min, respectively. Due to their great practical importance, AgNP-chitosan-silica composites can aspire to be classified as functional materials combining the environmental problem with microbiological activity.
Keywords: adsorption equilibrium; adsorption kinetics; biopolymer; chitosan; dye adsorption; nanosilver–chitosan–silica composite; silica.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures











References
-
- Sherafati Chaleshtori A., Marzhoseyni Z., Saeedi N., Azar Bahadori R., Mollazadeh S., Pourghadamyari H., Sajadimoghadam E., Abbaszadeh-Goudarzi K., Moradi Hasan-Abad A., Sharafati Chaleshtori R. Gelatin-based nanoparticles and antibiotics: A new therapeutic approach for osteomyelitis? Front. Mol. Biosci. 2024;11:1412325. doi: 10.3389/fmolb.2024.1412325. - DOI - PMC - PubMed
-
- Karachkovska A.D., Syrvatka V.J., Gromyko O.M., Fedorenko V.O. Nanoengineering: Fabrication, Properties, Optics, Thin Films, and Devices XVII. Volume 1146721 SPIE; Bellingham, WA, USA: 2020. Synthesis of antibiotic-silver nanoparticles complexes, their properties, and application for investigation of antibiotic-resistance mechanisms.
-
- Khurana C., Sharma P., Pandey O.P., Chudasama B. Synergistic Effect of Metal Nanoparticles on the Antimicrobial Activities of Antibiotics against Biorecycling Microbes. J. Mater. Sci. Technol. 2016;32:524–532. doi: 10.1016/j.jmst.2016.02.004. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

