Mucosal Bacterial Immunotherapy Attenuates the Development of Experimental Colitis by Reducing Inflammation Through the Regulation of Myeloid Cells
- PMID: 39769391
- PMCID: PMC11728189
- DOI: 10.3390/ijms252413629
Mucosal Bacterial Immunotherapy Attenuates the Development of Experimental Colitis by Reducing Inflammation Through the Regulation of Myeloid Cells
Abstract
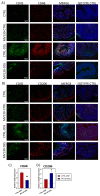
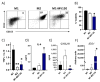
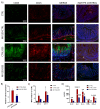
Ulcerative colitis is a chronic relapsing-remitting and potentially progressive form of inflammatory bowel disease in which there is extensive inflammation and mucosal damage in the colon and rectum as a result of an abnormal immune response. MV130 is a mucosal-trained immunity-based vaccine used to prevent respiratory tract infections in various clinical settings. Additionally, MV130 may induce innate immune cells that acquire anti-inflammatory properties and promote tolerance, which could have important implications for chronic inflammatory diseases such as ulcerative colitis. This work demonstrated that the prophylactic administration of MV130 substantially mitigated colitis in a mouse model of acute colitis induced by dextran sulphate sodium. MV130 downregulated systemic and local inflammatory responses, maintained the integrity of the intestinal barrier by preserving the enterocyte layer and goblet cells, and reduced the oedema and fibrosis characteristic of the disease. Mechanistically, MV130 significantly reduced the infiltration of neutrophils and pro-inflammatory macrophages in the intestinal wall of the diseased animals and favoured the appearance of M2-polarised macrophages. These results suggest that MV130 might have therapeutic potential for the treatment of ulcerative colitis, reducing the risk of relapse and the progression of disease.
Keywords: MV130; bacterial immunotherapy; immunomodulation; inflammation; macrophages; neutrophils; ulcerative colitis; vaccine.
Conflict of interest statement
J.L.S. is shareholder of Inmunotek S.L. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Shah S.C., Khalili H., Gower-Rousseau C., Olen O., Benchimol E.I., Lynge E., Nielsen K.R., Brassard P., Vutcovici M., Bitton A., et al. Sex-Based Differences in Incidence of Inflammatory Bowel Diseases-Pooled Analysis of Population-Based Studies from Western Countries. Gastroenterology. 2018;155:1079–1089.e3. doi: 10.1053/j.gastro.2018.06.043. - DOI - PubMed
MeSH terms
Substances
Grants and funding
- PID2021-123068OBI00/AEI/10.13039/501100011033/FEDER, UE/Spanish Ministry of Science and Innovation
- IND2020/BMD-17358/Council of Science University and innovation; Community of Madrid
- RD21/0017/0010/Instituto de Salud Carlos III co-funded by ERDF-Next Generation EU "Plan de Recuperación, Transformación y Resiliencia"
LinkOut - more resources
Full Text Sources

