In Silico Evaluation of Some Computer-Designed Fluoroquinolone-Glutamic Acid Hybrids as Potential Topoisomerase II Inhibitors with Anti-Cancer Effect
- PMID: 39770435
- PMCID: PMC11679884
- DOI: 10.3390/ph17121593
In Silico Evaluation of Some Computer-Designed Fluoroquinolone-Glutamic Acid Hybrids as Potential Topoisomerase II Inhibitors with Anti-Cancer Effect
Abstract
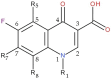
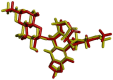
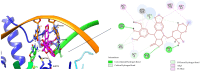
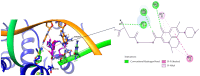
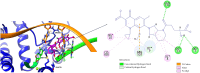
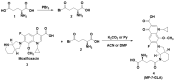
Background/Objectives: Fluoroquinolones (FQs) are topoisomerase II inhibitors with antibacterial activity, repositioned recently as anti-cancer agents. Glutamic acid (GLA) is an amino acid that affects human metabolism. Since an anti-cancer mechanism of FQs is human topoisomerase II inhibition, it is expected that FQ-GLA hybrids can act similarly. Methods: We designed 27 hypothetical hybrids of 6 FQs and GLA through amide bonds at the 3- and 7-position groups of FQs or via ethylenediamine/ethanolamine linkers at the carboxyl group of the FQ. Hydroxamic acid derivatives were also theoretically formulated. Computational methods were used to predict their physicochemical, pharmacokinetic, or toxicological properties and their anti-cancer activity. For comparison, etoposide was used as an anti-cancer agent inhibiting topoisomerase II. Molecular docking assessed whether the hybrids could interact with the human topoisomerase II beta in the same binding site and interaction sites as etoposide. Results: All the hybrids acted as potential topoisomerase II inhibitors, demonstrating possible anti-cancer activity on several cancer cell lines. Among all the proposed hybrids, MF-7-GLA would be the ideal candidate as a lead compound. The hybrid OF-3-EDA-GLA and the hydroxamic acid derivatives also stood out. Conclusions: Both FQs and GLA have advantageous structures for obtaining hybrids with favourable properties. Improvements in the hybrids' structure could lead to promising results.
Keywords: anti-cancer potential; computational methods; docking study; etoposide; fluoroquinolones; glutamic acid; huma topoisomerase II beta-DNA complex; hybrids.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
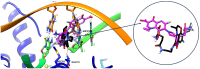
References
-
- Aranha O., Wood D.P., Sarkar F.H. Ciprofloxacin Mediated Cell Growth Inhibition, S/G2-M Cell Cycle Arrest, and Apoptosis in a Human Transitional Cell Carcinoma of the Bladder Cell Line. Clin. Cancer Res. 2000;6:891–900. - PubMed
-
- Perucca P., Savio M., Cazzalini O., Mocchi R., Maccario C., Sommatis S., Ferraro D., Pizzala R., Pretali L., Fasani E., et al. Structure–Activity Relationship and Role of Oxygen in the Potential Antitumour Activity of Fluoroquinolones in Human Epithelial Cancer Cells. J. Photochem. Photobiol. B Biol. 2014;140:57–68. doi: 10.1016/j.jphotobiol.2014.07.006. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

