Preclinical Study of a Dual-Target Molecular Probe Labeled with 68Ga Targeting SSTR2 and FAP
- PMID: 39770488
- PMCID: PMC11677724
- DOI: 10.3390/ph17121647
Preclinical Study of a Dual-Target Molecular Probe Labeled with 68Ga Targeting SSTR2 and FAP
Abstract
Objective: Currently, 68Ga-labeled somatostatin analogs (SSAs) are the most commonly used imaging agents for patients with neuroendocrine tumors (NETs) in clinical practice, demonstrating good results in tumor diagnosis. For applications in peptide receptor radionuclide therapy (PRRT), targeted drugs should have high tumor uptake and prolonged tumor retention time. To enhance the uptake and retention of tracers in NETs, our goal is to design a 68Ga-labeled heterodimer for optimizing pharmacokinetics and assess whether this form is more efficacious than its monomeric equivalents.
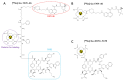
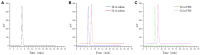
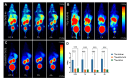
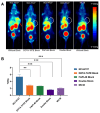
Methods: Using the somatostatin analog TATE and quinoline-based compound FAPI-46 as raw materials, we designed and synthesized 68Ga-labeled TATE-46. The labeling efficiency and stability were verified by Radio-HPLC. The receptor binding properties and tumor targeting were examined both in vitro and in vivo by using NCI-H727 (SSTR2/FAP, positive) and Mc38 (SSTR2/FAP, negative) cell lines and tumor-bearing mouse models. Preclinical evaluation was performed through cell uptake, pharmacokinetics, Micro PET, and biodistribution studies, and the results were compared with [68Ga]Ga-DOTA-TATE and [68Ga]Ga -FAPI-46. Immunohistochemistry and HE staining were performed on tumor tissues from tumor-bearing mice for further validation.
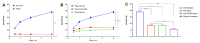
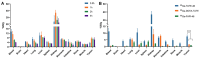
Results: [68Ga]Ga-TATE-46 showed comparable SSTR2 and FAP targeting ability to monomeric TATE and FAPI-46 in cell uptake and PET imaging studies. [68Ga]Ga-TATE-46 exhibited significantly higher uptake in NCI-H727 (SSTR2/FAP, positive) tumors compared to [68Ga]Ga-DOTA-TATE (p < 0.001) and [68Ga]Ga-FAPI-46 (p < 0.001). No increased uptake of [68Ga]Ga-TATE-46 was observed in MC38 tumors (SSTR2/FAP, negative). Additionally, excess DOTA-TATE and/or unlabeled FAPI-46 significantly blocked the uptake of [68Ga]Ga-TATE-46 in NCI-H727 tumors (p < 0.001), confirming its dual-receptor targeting characteristics. The ex vivo biodistribution, immunofluorescence and immunohistochemistry results were in line with the in vivo imaging findings.
Conclusion: Compared with 68Ga-labeled FAPI-46 and DOTA-TATE mono-specific tracers, the dual-target tracer [68Ga]Ga-TATE-46 improves tumor uptake, extends tumor retention, and enhances pharmacokinetics. It is an effective probe for non-invasive detection of tumors expressing FAP and SSTR2, and it is worth further studying its application in the expression of sstr2 and FAP-related tumors.
Keywords: PET; fibroblast activation protein; heterodimer; somatostatin receptor 2.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Mizutani G., Nakanishi Y., Watanabe N., Honma T., Obana Y., Seki T., Ohni S., Nemoto N. Expression of Somatostatin Receptor (SSTR) Subtypes (SSTR-1, 2A, 3, 4 and 5) in Neuroendocrine Tumors Using Real-time RT-PCR Method and Immunohistochemistry. Acta Histochem. ET Cytochem. 2012;45:167–176. doi: 10.1267/ahc.12006. - DOI - PMC - PubMed
-
- Hope T.A., Allen-Auerbach M., Bodei L., Calais J., Dahlbom M., Dunnwald L.K., Graham M.M., Jacene H.A., Heath C.L., Mittra E.S., et al. SNMMI Procedure Standard/EANM Practice Guideline for SSTR PET: Imaging Neuroendocrine Tumors. J. Nucl. Med. 2023;64:204–210. doi: 10.2967/jnumed.122.264860. - DOI - PubMed
-
- Zhao L., Pang Y., Fang J., Chen J., Zhou Y., Sun L., Wu H., Guo Z., Lin Q., Chen H. Design, Preclinical Evaluation, and Clinical Translation of 68Ga-FAPI-LM3, a Heterobivalent Molecule for PET Imaging of Nasopharyngeal Carcinoma. J. Nucl. Med. 2024;65:394–401. doi: 10.2967/jnumed.123.266183. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

