Antinociceptive Potential of Ximenia americana L. Bark Extract and Caffeic Acid: Insights into Pain Modulation Pathways
- PMID: 39770512
- PMCID: PMC11677608
- DOI: 10.3390/ph17121671
Antinociceptive Potential of Ximenia americana L. Bark Extract and Caffeic Acid: Insights into Pain Modulation Pathways
Abstract
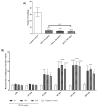
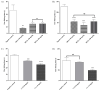
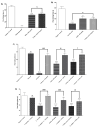
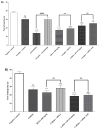
Background/Objectives: This study evaluated the antinociceptive effect of the Ximenia americana L. bark extract (HEXA) and its primary component, caffeic acid (CA), through in vivo assays. Methods: The antinociceptive properties were assessed using abdominal writhing, hot plate, and Von Frey tests. Additionally, the study investigated the modulation of various pain signaling pathways using a pharmacological approach. Results: The results demonstrated that all doses of the HEXA significantly increased latency in the hot plate test, decreased the number of abdominal contortions, reduced hyperalgesia in the Von Frey test, and reduced both phases of the formalin test. Caffeic acid reduced licking time in the first phase of the formalin test at all doses, with the highest dose showing significant effects in the second phase. The HEXA potentially modulated α2-adrenergic (52.99%), nitric oxide (57.77%), glutamatergic (33.66%), vanilloid (39.84%), cyclic guanosine monophosphate (56.11%), and K+ATP channel-dependent pathways (38.70%). Conversely, CA influenced the opioid, glutamatergic (53.60%), and vanilloid (34.42%) pathways while inhibiting nitric oxide (52.99%) and cyclic guanosine monophosphate (38.98%). Conclusions: HEXA and CA exhibit significant antinociceptive effects due to their potential interference in multiple pain signaling pathways. While the molecular targets remain to be fully investigated, HEXA and CA demonstrate significant potential for the development of new analgesic drugs.
Keywords: HPLC; anti-inflammatory; mechanism of action; natural compounds; nociception; pain.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




Similar articles
-
The leaves of Bougainvillea spectabilis suppressed inflammation and nociception in vivo through the modulation of glutamatergic, cGMP, and ATP-sensitive K+ channel pathways.J Ethnopharmacol. 2020 Oct 28;261:113148. doi: 10.1016/j.jep.2020.113148. Epub 2020 Jul 18. J Ethnopharmacol. 2020. PMID: 32687959
-
Root bark of Discaria americana attenuates pain: A pharmacological evidence of interaction with opioidergic system and TRP/ASIC channels.J Ethnopharmacol. 2018 Dec 5;227:258-267. doi: 10.1016/j.jep.2018.09.008. Epub 2018 Sep 7. J Ethnopharmacol. 2018. PMID: 30201229
-
Antinociceptive effect of tadalafil in various pain models: Involvement of opioid receptors and nitric oxide cyclic GMP pathway.Toxicol Appl Pharmacol. 2018 Aug 1;352:170-175. doi: 10.1016/j.taap.2018.05.013. Epub 2018 May 17. Toxicol Appl Pharmacol. 2018. PMID: 29777732
-
Lysionotin exerts antinociceptive effects in various models of nociception induction.Heliyon. 2023 Apr 18;9(4):e15619. doi: 10.1016/j.heliyon.2023.e15619. eCollection 2023 Apr. Heliyon. 2023. PMID: 37151635 Free PMC article.
-
Central antinociceptive effects of non-steroidal anti-inflammatory drugs and paracetamol. Experimental studies in the rat.Acta Anaesthesiol Scand Suppl. 1995;103:1-44. Acta Anaesthesiol Scand Suppl. 1995. PMID: 7725891 Review.
References
LinkOut - more resources
Full Text Sources

