Centella asiatica Promotes Antioxidant Gene Expression and Mitochondrial Oxidative Respiration in Experimental Autoimmune Encephalomyelitis
- PMID: 39770523
- PMCID: PMC11676818
- DOI: 10.3390/ph17121681
Centella asiatica Promotes Antioxidant Gene Expression and Mitochondrial Oxidative Respiration in Experimental Autoimmune Encephalomyelitis
Abstract
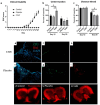
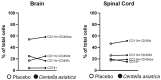
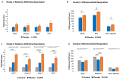
Background/Objectives:Centella asiatica (L.) Urban (family Apiaceae) (C. asiatica) is a traditional botanical medicine used in aging and dementia. Water extracts of C. asiatica (CAW) have been used to treat neuropsychiatric symptoms in related animal models and are associated with increases in antioxidant response element (ARE) genes and improvements in mitochondrial respiratory function and neuronal health. Because multiple sclerosis (MS) shares its neurogenerative pathology of oxidative stress and mitochondrial dysfunction with aging and dementia, neuropsychiatric symptoms in MS may also benefit from C. asiatica. To determine whether CAW similarly benefits neuropsychiatric symptoms, ARE gene expression, and mitochondrial respiration in inflammatory models of MS, and to determine the effects of CAW on clinical disability and inflammation, we tested CAW using experimental autoimmune encephalomyelitis (EAE). Methods: C57BL/6J mice induced with EAE were treated with CAW or a placebo for 2 weeks. The outcomes were clinical disability, signs of anxiety (open field test), ARE gene expression, mitochondrial respiration, and inflammation and demyelination. Results: At the dosing schedule and concentrations tested, CAW-treated mice with EAE demonstrated increased ARE gene expression and mitochondrial respiratory activity compared to those of placebo-treated mice with EAE. CAW was also associated with reduced inflammatory infiltrates in the spinal cord, but the differences between the populations of activated versus quiescent microglia were equivocal. CAW did not improve behavioral performance, EAE motor disability, or demyelination. Conclusions: In the inflammatory EAE model of MS, CAW demonstrates similar neuroprotective effects to those it exhibits in aging and dementia mouse models. These benefits, along with the anti-inflammatory effects of CAW, support further investigation of its neuropsychiatric effects in people with MS.
Keywords: Centella asiatica; antioxidant; experimental autoimmune encephalomyelitis; mitochondrial respiration.
Conflict of interest statement
Author Payel Kundu is employed by the company Stantec ChemRisk but was not employed by this company at the time of conducting the experiments. Dr. Kundu and the remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Update of
-
Centella asiatica promotes antioxidant gene expression and mitochondrial oxidative respiration in experimental autoimmune encephalomyelitis.Res Sq [Preprint]. 2023 Oct 6:rs.3.rs-3393042. doi: 10.21203/rs.3.rs-3393042/v1. Res Sq. 2023. Update in: Pharmaceuticals (Basel). 2024 Dec 13;17(12):1681. doi: 10.3390/ph17121681. PMID: 37886497 Free PMC article. Updated. Preprint.
Similar articles
-
Centella asiatica promotes antioxidant gene expression and mitochondrial oxidative respiration in experimental autoimmune encephalomyelitis.Res Sq [Preprint]. 2023 Oct 6:rs.3.rs-3393042. doi: 10.21203/rs.3.rs-3393042/v1. Res Sq. 2023. Update in: Pharmaceuticals (Basel). 2024 Dec 13;17(12):1681. doi: 10.3390/ph17121681. PMID: 37886497 Free PMC article. Updated. Preprint.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Galantamine for Alzheimer's disease.Cochrane Database Syst Rev. 2002;(3):CD001747. doi: 10.1002/14651858.CD001747. Cochrane Database Syst Rev. 2002. Update in: Cochrane Database Syst Rev. 2004 Oct 18;(4):CD001747. doi: 10.1002/14651858.CD001747.pub2. PMID: 12137632 Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Cell therapy procedure using anti-inflammatory macrophage M2 can potentially reduce Clinical Score in animals with Experimental Autoimmune Encephalomyelitis: A preclinical systematic review and meta-analysis study.Fundam Clin Pharmacol. 2023 Apr;37(2):215-225. doi: 10.1111/fcp.12844. Epub 2022 Nov 14. Fundam Clin Pharmacol. 2023. PMID: 36300567
Cited by
-
In Vitro Neuroprotective Effects of a Mixed Extract of Bilberry, Centella asiatica, Hericium erinaceus, and Palmitoylethanolamide.Foods. 2025 Jul 30;14(15):2678. doi: 10.3390/foods14152678. Foods. 2025. PMID: 40807615 Free PMC article.
References
-
- Rommer P.S., Eichstädt K., Ellenberger D., Flachenecker P., Friede T., Haas J., Kleinschnitz C., Pöhlau D., Rienhoff O., Stahmann A., et al. Symptomatology and symptomatic treatment in multiple sclerosis: Results from a nationwide MS registry. Mult. Scler. 2019;25:1641–1652. doi: 10.1177/1352458518799580. - DOI - PubMed
-
- Krupp L.B., Christodoulou C., Melville P., Scherl W.F., Pai L.Y., Muenz L.R., He D., Benedict R.H., Goodman A., Rizvi S., et al. Multicenter randomized clinical trial of donepezil for memory impairment in multiple sclerosis. Neurology. 2011;76:1500–1507. doi: 10.1212/WNL.0b013e318218107a. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

