Simple and Efficient Synthesis of N-Succinimidyl-4-[18F]fluorobenzoate ([18F]SFB)-An Important Intermediate for the Introduction of Fluorine-18 into Complex Bioactive Compounds
- PMID: 39770565
- PMCID: PMC11677304
- DOI: 10.3390/ph17121723
Simple and Efficient Synthesis of N-Succinimidyl-4-[18F]fluorobenzoate ([18F]SFB)-An Important Intermediate for the Introduction of Fluorine-18 into Complex Bioactive Compounds
Abstract
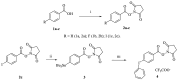
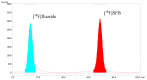
Background:N-succinimidyl-[18F]fluorobenzoate ([18F]SFB) is commonly prepared through a three-step procedure starting from [18F]fluoride ion. A number of methods for the single-step radiosynthesis of [18F]SFB have been introduced recently, including the radiofluorination of diaryliodonium salts and the Cu-mediated 18F-fluorination of pinacol aryl boronates and aryl tributyl stannanes, but they still have the drawbacks of lengthy product purification procedures. In the present work, two approaches for the direct labeling of [18F]SFB from diaryliodonium (DAI) salt (4) and pinacol aryl boronate (6) are evaluated, with a major focus on developing a fast and simple SPE-based purification procedure. Methods: DAI salt precursor 6 was labeled employing the common "minimalist" approach with a two-step reaction heating sequence. The Cu-mediated radiofluorination of 4 was accomplished using Bu4NOTf as a phase transfer catalyst for the elution of [18F]fluoride, followed by radiofluorination in the same solvent. Several types of SPE cartridges were tested in the elution and SPE procedures. Results: The Cu-mediated 18F-fluorination of the pinacol aryl boronate precursor afforded a higher RCC of 56 ± 3% (n = 7), making it better suited for the one-pot synthesis of [18F]SFB. SPE-based purification was achieved using cation exchange and reverse-phase polymer resin cartridges, connected in series. In a full-batch test, [18F]SFB was obtained with an RCY of 30% (n. d. c.), RCP > 99%, Am 96-155 GBq/µmol, and a synthesis time of ≤35 min. Conclusions: Compared to other published methods, [18F]SFB production via the Cu-mediated radiofluorination of pinacol aryl boronate precursor provides significant time and cost savings, coupled with an ease of implementation.
Keywords: [18F]SFB; cartridge-based purification; copper-mediated radiofluorination; fluorine-18; one-step synthesis; prosthetic group.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
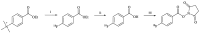


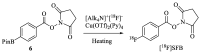
References
LinkOut - more resources
Full Text Sources

