The Significant Therapeutic Effects of Chinese Scorpion: Modern Scientific Exploration of Ion Channels
- PMID: 39770577
- PMCID: PMC11678150
- DOI: 10.3390/ph17121735
The Significant Therapeutic Effects of Chinese Scorpion: Modern Scientific Exploration of Ion Channels
Abstract
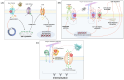
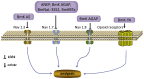
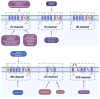
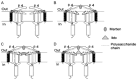
Chinese scorpion (CS), a traditional animal-based medicine used for over a millennium, has been documented since AD 935-960. It is derived from the scorpion Buthus martensii Karsch and is used to treat various ailments such as stroke, epilepsy, rheumatism, and more. Modern research has identified the pharmacological mechanisms behind its traditional uses, with active components like venom and proteins showing analgesic, antitumor, antiepileptic, and antithrombotic effects. Studies reveal that CS affects ion channels, crucial for cellular functions, through interactions with sodium, potassium, and calcium channels, potentially explaining its therapeutic effects. Future research aims to elucidate the precise mechanisms, target specific ion channel subtypes, and validate clinical efficacy and safety, paving the way for novel therapies based on these natural compounds.
Keywords: Buthus martensii Karsch; chemical components; ion channel; pharmacological effects.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Liang X., Yuan H., Yang Y., Wang E., Ma C., Zhan Z. Herbal textual research on Scorpio in famous classical formulas and its resource status. Chin. J. Exp. Tradit. Med. Formulae. 2023;29:34–43. doi: 10.13422/j.cnki.syfjx.20220746. - DOI
-
- Yang Z., Ji L., Liu H., Bian S., Xu G., Lv S. Research progress the study of chemical constituents and pharmacological effects of scorpion. Cent. South Pharm. 2020;18:1523–1529.
-
- Qin C. Ph.D. Thesis. Wuhan University; Wuhan, China: 2021. Research on Discovery, Function and Mechanism of Potassium Channel-Inhibiting Peptides in Processed Scorpion Medicinal Material in Traditional Chinese Medicine.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

