Gut Microbiota Comparison in Rectal Swabs Versus Stool Samples in Cats with Kidney Stones
- PMID: 39770613
- PMCID: PMC11677927
- DOI: 10.3390/microorganisms12122411
Gut Microbiota Comparison in Rectal Swabs Versus Stool Samples in Cats with Kidney Stones
Abstract
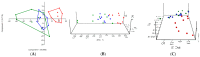
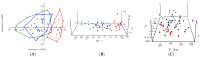
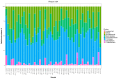
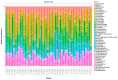
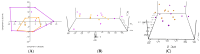
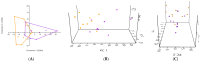
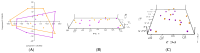
To investigate the role of the intestinal bacterial microbiota in the pathogenesis of calcium oxalate nephrolithiasis in cats, a condition characterized by the formation of kidney stones, it is desirable to identify a sample collection method that accurately reflects the microbiota's composition. The objective of this study was to evaluate the impact of fecal sample collection methods on the intestinal microbiota composition in two cat populations: healthy cats and kidney stone-diseased cats. The study included eighteen cats from the same colony, comprising nine healthy cats and nine cats with spontaneously occurring presumed calcium oxalate kidney stones. Three fecal collection methods were compared: rectal swabs, the collection of fresh stool, and the collection of stool exposed to ambient air for 24 h. The bacterial microbiota was analyzed through the high-resolution sequencing of the V3-V4 region of the 16S rRNA gene. For all cats, within the same individual, a one-way PERMANOVA analysis showed a significant difference between the rectal swabs and fresh stool (p = 0.0003), as well as between the rectal swabs and stool exposed to ambient air for 24 h (p = 0.0003), but no significant difference was identified between the fresh stool and non-fresh stool (p = 0.0651). When comparing the two populations of cats, this study provides seemingly conflicting results. (1) A principal component analysis (PCA) comparison revealed a significant difference in the bacterial composition between the healthy cats and the cats with kidney stones only when the sample was a fresh fecal sample (p = 0.0037). This finding suggests that the intestinal bacteria involved in the pathogenesis of kidney stones in cats are luminal and strictly anaerobic bacteria. Consequently, exposure to ambient air results in a loss of information, preventing the identification of dysbiosis. For clinical studies, non-fresh stool samples provided by owners does not appear suitable for studying the gut microbiota of cats with kidney stones; fresh stool should be favored. (2) Interestingly, the rectal swabs alone highlighted significant differences in the proportion of major phyla between the two populations. These findings highlight the critical importance of carefully selecting fecal collection methods when studying feline gut microbiota. Combining rectal swabs and fresh stool sampling provides complementary insights, offering the most accurate understanding of the gut microbiota composition in the context of feline kidney stone pathogenesis.
Keywords: calcium oxalate; cat; kidney stones; lithiasis; microbiota; renal calculi.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

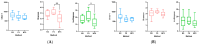
References
-
- Lin E., Xu J., Liu M., Nazzal L., Katz S. Enteric Hyperoxaluria and Kidney Stone Management in Inflammatory Bowel Disease. Curr. Treat. Options Gastroenterol. 2020;18:384–393. doi: 10.1007/s11938-020-00295-x. - DOI
-
- Kachroo N., Lange D., Penniston K.L., Stern J., Tasian G., Bajic P., Wolfe A.J., Suryavanshi M., Ticinesi A., Meschi T., et al. Meta-analysis of Clinical Microbiome Studies in Urolithiasis Reveal Age, Stone Composition, and Study Location as the Predominant Factors in Urolithiasis-Associated Microbiome Composition. mBio. 2021;12:e0200721. doi: 10.1128/mBio.02007-21. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

