Neurotransmitter Metabolic Disturbance in Methamphetamine Abusers: Focus on Tryptophan and Tyrosine Metabolic Pathways
- PMID: 39771127
- PMCID: PMC11728700
- DOI: 10.3390/toxics12120912
Neurotransmitter Metabolic Disturbance in Methamphetamine Abusers: Focus on Tryptophan and Tyrosine Metabolic Pathways
Abstract
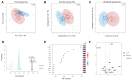
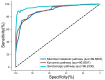
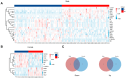
Methamphetamine (METH) abuse disrupts the homeostasis of neurotransmitter (NT) metabolism, contributing to a wide range of neurological and psychological disorders. However, the specific effects of METH on NT metabolism, particularly for the tryptophan (TRP) and tyrosine (TYR) metabolic pathways, remain poorly understood. In this study, serum samples from 78 METH abusers and 79 healthy controls were analyzed using Ultra-High-Performance Liquid Chromatography with Tandem Mass Spectrometry (UHPLC-MS/MS). A total of 41 substances, primarily from the TRP and TYR metabolic pathways, were detected and subjected to multivariate analysis. Principal Component Analysis (PCA) and Partial Least Squares Discriminant Analysis (PLS-DA) revealed a significant separation of serum metabolites between METH abusers and controls, encompassing the disturbance of serotonergic, kynurenic, and microbial metabolism. In the serotonergic pathway, METH significantly reduced melatonin (MLT) levels and impaired the conversion of serotonin (5-HT) to N-acetylserotonin (NAS), a key precursor of MLT. In the kynurenic pathway, METH promoted a shift to the toxic metabolic pathway, evidenced by elevated levels of 3-hydroxykynurenine (3-HK) and quinolinic acid (QA). Furthermore, microbial metabolic pathway-related indole and its derivatives were markedly suppressed in METH abusers. Gender-specific differences were also observed, with NT metabolism in TRP and TYR pathways showing more pronounced alterations in male or female subgroups. Therefore, the current study provides a comprehensive overview of the disturbance in TRP- and TYR-associated NT metabolism caused by METH abuse and highlights NT metabolism as a promising therapeutic target for METH-induced neural and psychiatric disorders.
Keywords: methamphetamine; neurotransmitters; tryptophan metabolism; tyrosine metabolism.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures




References
-
- Zhang K., Chen L., Yang J., Liu J., Li J., Liu Y., Li X., Chen L., Hsu C., Zeng J., et al. Gut Microbiota-Derived Short-Chain Fatty Acids Ameliorate Methamphetamine-Induced Depression- and Anxiety-like Behaviors in a Sigmar-1 Receptor-Dependent Manner. Acta Pharm. Sin. B. 2023;13:4801–4822. doi: 10.1016/j.apsb.2023.09.010. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

