Improvement of End-of-Synthesis Radiochemical Purity of 177Lu-DOTA-PSMA-Ligands with Alternative Synthesis Approaches: Conversion Upswing and Side-Products Minimization
- PMID: 39771514
- PMCID: PMC11677419
- DOI: 10.3390/pharmaceutics16121535
Improvement of End-of-Synthesis Radiochemical Purity of 177Lu-DOTA-PSMA-Ligands with Alternative Synthesis Approaches: Conversion Upswing and Side-Products Minimization
Abstract
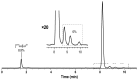
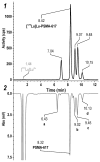
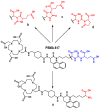
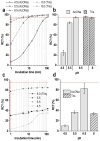
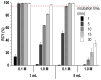
Background: Radiochemical purity is a key criterion for the quality of radiopharmaceuticals used in clinical practice. The joint improvement of analytical methods capable of identifying related radiochemical impurities and determining the actual radiochemical purity, as well as the improvement of synthesis methods to minimize the formation of possible radiochemical impurities, is integral to the implementation of high-tech nuclear medicine procedures. PSMA-targeted radionuclide therapy with lutetium-177 has emerged as an effective treatment option for prostate cancer, and [177Lu]Lu-PSMA-617 and [177Lu]Lu-PSMAI&T have achieved global recognition as viable radiopharmaceuticals. Recently, it was shown that specific radiochemical impurities can form during the synthesis of [177Lu]Lu-PSMA-617 because of a spontaneous, thermally mediated condensation of the Glu-C(O)-Lys fragment, resulting in the formation of three different cyclic forms (with no affinity for PSMA). During this study, we identified another impurity, a product of detachment of the Glu-CO fragment from PSMA-617, caused by heating. The total content of all four thermally mediated degradation products may reach 9-11% during classical incubation for 30 min at 95 °C, reducing the radiochemical purity to an unacceptable level (albeit with high levels of radiochemical conversion). It is reasonable to assume that the formation of similar impurities is characteristic of all PSMA-specific vectors that contain Glu-C(O)-Lys pharmacophores. Because the formation of these impurities directly depends on the temperature and incubation time, to reduce their content in the reaction mixture at the end of the synthesis, it is necessary to select conditions to achieve a high level of radiochemical conversion for the minimum possible time and/or at the minimum sufficient temperature.
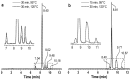
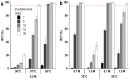
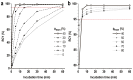
Methods: In this study, using [177Lu]Lu-PSMA-617 as an example, we evaluated the efficiency of alternative methods of synthesis with microwave heating and co-solvent (ethanol) addition to ensure radiochemical yield and radiochemical purity in the shortest possible time and at the minimum necessary and sufficient synthesis temperature.
Results: Both approaches achieved a significant reduction in the impurities content, while achieving satisfactory synthesis yields in a short time. In addition to improving the synthesis parameters and radiochemical purity, the use of microwave heating and the addition of ethanol reduces the negative influence of other auxiliaries on labeling kinetics. Notably, the addition of ethanol under certain conditions allowed [177Lu]Lu-PSMA-617 to be synthesized at room temperature for only 10 min. This makes it possible to achieve exceptionally high real radiochemical purity of the preparations, determined only by the quality of the original precursor. The approaches considered in this study can be successfully applied to improve the synthesis process and quality parameters of the finished product, both for known radiopharmaceuticals and for those under development.
Keywords: DOTA; PSMA; [177Lu]Lu-PSMA-617; ethanol; lutetium-177; microwave heating; mixed media; radiochemical purity; radiochemical yield; related impurities.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures






Similar articles
-
Identification, Characterization, and Suppression of Side Products Formed during the Synthesis of [177Lu]Lu-PSMA-617.J Med Chem. 2021 Apr 22;64(8):4960-4971. doi: 10.1021/acs.jmedchem.1c00045. Epub 2021 Apr 7. J Med Chem. 2021. PMID: 33826320
-
Radiolysis-Associated Decrease in Radiochemical Purity of 177Lu-Radiopharmaceuticals and Comparison of the Effectiveness of Selected Quenchers against This Process.Molecules. 2023 Feb 16;28(4):1884. doi: 10.3390/molecules28041884. Molecules. 2023. PMID: 36838872 Free PMC article.
-
The development of 177Lu-DOTA-CC-PSMA following a unified "Click Chemistry" protocol of synthesizing metal nuclide-conjugated radiopharmaceuticals.EJNMMI Radiopharm Chem. 2024 Jul 31;9(1):56. doi: 10.1186/s41181-024-00287-7. EJNMMI Radiopharm Chem. 2024. PMID: 39083135 Free PMC article.
-
Synthesis of 68Ga-radiopharmaceuticals using both generator-derived and cyclotron-produced 68Ga as exemplified by [68Ga]Ga-PSMA-11 for prostate cancer PET imaging.Nat Protoc. 2022 Apr;17(4):980-1003. doi: 10.1038/s41596-021-00662-7. Epub 2022 Mar 4. Nat Protoc. 2022. PMID: 35246649 Review.
-
Towards Improving the Efficacy of PSMA-Targeting Radionuclide Therapy for Late-Stage Prostate Cancer-Combination Strategies.Curr Oncol Rep. 2023 Nov;25(11):1363-1374. doi: 10.1007/s11912-023-01458-6. Epub 2023 Oct 20. Curr Oncol Rep. 2023. PMID: 37861915 Free PMC article. Review.
References
-
- Burkett B.J., Bartlett D.J., McGarrah P.W., Lewis A.R., Johnson D.R., Berberoğlu K., Pandey M.K., Packard A.T., Halfdanarson T.R., Hruska C.B., et al. A Review of Theranostics: Perspectives on Emerging Approaches and Clinical Advancements. Radiol. Imaging Cancer. 2023;5:e220157. doi: 10.1148/rycan.220157. - DOI - PMC - PubMed
-
- Lapi S.E., Scott P.J.H., Scott A.M., Windhorst A.D., Zeglis B.M., Abdel-Wahab M., Baum R.P., Buatti J.M., Giammarile F., Kiess A.P., et al. Recent Advances and Impending Challenges for the Radiopharmaceutical Sciences in Oncology. Lancet Oncol. 2024;25:e236–e249. doi: 10.1016/S1470-2045(24)00030-5. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

