Evaluating the risk of acute kidney injury and mortality associated with concomitant use of vancomycin with piperacillin/tazobactam or meropenem in critically ill and non-critically ill patients: a systematic review and meta-analysis
- PMID: 39773457
- PMCID: PMC11706157
- DOI: 10.1186/s12879-024-10227-0
Evaluating the risk of acute kidney injury and mortality associated with concomitant use of vancomycin with piperacillin/tazobactam or meropenem in critically ill and non-critically ill patients: a systematic review and meta-analysis
Abstract
Background: There are conflicting findings regarding the risk of acute kidney injury (AKI) and mortality with vancomycin/piperacillin-tazobactam combination (VPT) and vancomycin/meropenem (VM). The aim of this meta-analysis was to compare the risk of AKI and mortality between VPT and VM.
Methods: Observational studies reporting the incidence of AKI and mortality in patients receiving VPT or VM between January 2017 and September 2024 were retrieved from PubMed, the Cochrane Library, and Web of Science. The primary outcome of the analysis was the risk of AKI, and the secondary outcomes were the mortality rate, need for renal replacement therapy (RRT), and hospital length of stay (LOS). This meta-analysis was conducted using a random-effects model to estimate the odds ratios (OR) and 95% confidence intervals (CI) for AKI, mortality, and RRT or mean difference and 95% CI for the LOS.
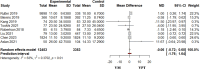
Results: Seventeen studies involving a total of 80,595 patients were included in the analysis. The odds of developing AKI were higher among patients who received the VPT versus those who received the VM combination (OR = 2.02; 95%CI 1.56-2.62). There were no differences between VPT and VM in the mortality and hospital length of stay; however, the odds of requiring RRT were higher among VPT group versus VM group (OR = 1.55; 95%CI 1.23-1.96).
Conclusion: The findings suggest that the use of VPT is associated with a higher risk of AKI compared to VM and highlight the need for cautious antibiotic selection and monitoring of renal function in patients receiving these combinations.
Keywords: Acute kidney injury; Beta-lactams; Meropenem; Nephrotoxicity; Piperacillin-tazobactam; Vancomycin.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Competing interests: The authors declare no competing interests.
Figures



Similar articles
-
Vancomycin combined with piperacillin/tazobactam increases the risk of acute kidney injury compared with vancomycin plus other anti-pseudomonal beta-lactams: a systematic review and network meta-analysis.J Antimicrob Chemother. 2025 Jan 3;80(1):47-58. doi: 10.1093/jac/dkae410. J Antimicrob Chemother. 2025. PMID: 39533846
-
Incidence of Acute Kidney Injury Among Patients Receiving the Combination of Vancomycin with Piperacillin-Tazobactam or Meropenem.Pharmacotherapy. 2018 Dec;38(12):1184-1193. doi: 10.1002/phar.2179. Epub 2018 Oct 3. Pharmacotherapy. 2018. PMID: 30175410
-
Nephrotoxic Risk Associated With Combination Therapy of Vancomycin and Piperacillin-Tazobactam in Critically Ill Patients With Chronic Kidney Disease.J Intensive Care Med. 2024 Sep;39(9):860-865. doi: 10.1177/08850666241234577. Epub 2024 Feb 28. J Intensive Care Med. 2024. PMID: 38415281
-
Incidence of Acute Kidney Injury in Critically Ill Patients Receiving Vancomycin with Concomitant Piperacillin-Tazobactam, Cefepime, or Meropenem.Antimicrob Agents Chemother. 2019 Apr 25;63(5):e02658-18. doi: 10.1128/AAC.02658-18. Print 2019 May. Antimicrob Agents Chemother. 2019. PMID: 30782987 Free PMC article.
-
Epidemiology of Vancomycin in Combination With Piperacillin/Tazobactam-Associated Acute Kidney Injury in Children: A Systematic Review and Meta-analysis.Ann Pharmacother. 2024 Oct;58(10):1034-1044. doi: 10.1177/10600280231220379. Epub 2024 Jan 27. Ann Pharmacother. 2024. PMID: 38279799
References
-
- Levey A.S. Defining AKD: the Spectrum of AKI, AKD, and CKD. Nephron. 2022;146(3):302–5. 10.1159/000516647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

