Trajectories of Change, Illness Understanding, and Parental Worries in Children and Adolescents Undergoing Internet-Delivered Cognitive-Behavioral Therapy for Functional Abdominal Pain Disorders: Protocol for a Single-Case Design and Explorative Pilot Study
- PMID: 39773759
- PMCID: PMC11751652
- DOI: 10.2196/58563
Trajectories of Change, Illness Understanding, and Parental Worries in Children and Adolescents Undergoing Internet-Delivered Cognitive-Behavioral Therapy for Functional Abdominal Pain Disorders: Protocol for a Single-Case Design and Explorative Pilot Study
Abstract
Background: Functional abdominal pain disorders (FAPDs) are common in young people and are characterized by persistent or recurrent abdominal symptoms without apparent structural or biochemical abnormalities. FAPDs are associated with diminished quality of life, school absence, increased health care use, and comorbid anxiety and depression. Exposure-based internet-delivered cognitive behavioral therapy (ICBT) has demonstrated efficacy in alleviating abdominal symptoms and improving quality of life. However, a deeper understanding of effect mechanisms and identification of possible additional treatment targets could refine treatment.
Objective: This protocol paper aims to describe a study focusing on children and adolescents undergoing ICBT for FAPDs, aiming to further investigate the underlying mechanisms of effect.
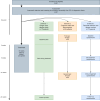
Methods: Children (8-12 years), adolescents (13-17 years) with FAPDs, and their respective parents will be included for 10 weeks for ICBT. First, detailed trajectories of effect are examined through a randomized single-case design study involving 6 children and 6 adolescents (substudy 1). Following this, an open-ended explorative pilot study with 30 children and 30 adolescents explores potential illness-related cognitive biases and interoceptive accuracy before and after treatment (substudy 2). Finally, spanning across these 2 substudies, including all parents from substudies 1 and 2, we will assess parental distress and illness worries before and after treatment, and how these factors impact the treatment adherence and outcomes of the child or adolescent (substudy 3).
Results: Recruitment of participants began in June 2022 and is finalized for substudy 1 and ongoing for substudies 2 and 3. Recruitment is expected to be completed by January 2025, with final data collection during April 2025.
Conclusions: The findings have the potential to contribute to the ongoing improvement of specialized psychological treatment for FAPDs in young people.
Trial registration: ClinicalTrials.gov NCT05237882; https://clinicaltrials.gov/study/NCT05237882; ClinicalTrials.gov NCT05486585; https://clinicaltrials.gov/study/NCT05486585; OSF Registries osf.io/c49k7; https://osf.io/c49k7.
International registered report identifier (irrid): DERR1-10.2196/58563.
Keywords: abdominal pain; adolescents; attentional bias; children; cognitive behavioral therapy; functional abdominal pain disorders; internet-based intervention; interoception; medication; parental distress; psychological treatment; psychology; quality of life; single case study; study protocol; treatment; youth.
©Eva Skovslund Nielsen, Karen Kallesøe, Tine Bennedsen Gehrt, Ellen Bjerre-Nielsen, Maria Lalouni, Lisbeth Frostholm, Marianne Bonnert, Charlotte Ulrikka Rask. Originally published in JMIR Research Protocols (https://www.researchprotocols.org), 07.01.2025.
Conflict of interest statement
Conflicts of Interest: None declared.
Figures

References
-
- Boronat AC, Ferreira-Maia AP, Matijasevich A, Wang YP. Epidemiology of functional gastrointestinal disorders in children and adolescents: a systematic review. World J Gastroenterol. 2017;23(21):3915–3927. doi: 10.3748/wjg.v23.i21.3915. https://www.wjgnet.com/1007-9327/full/v23/i21/3915.htm - DOI - PMC - PubMed
-
- Robin SG, Keller C, Zwiener R, Hyman PE, Nurko S, Saps M, Di Lorenzo C, Shulman RJ, Hyams JS, Palsson O, van Tilburg MAL. Prevalence of pediatric functional gastrointestinal disorders utilizing the Rome IV criteria. J Pediatr. 2018;195:134–139. doi: 10.1016/j.jpeds.2017.12.012.S0022-3476(17)31634-7 - DOI - PubMed
-
- Varni JW, Bendo CB, Nurko S, Shulman RJ, Self MM, Franciosi JP, Saps M, Pohl JF, Pediatric Quality of Life Inventory (PedsQL) Gastrointestinal Symptoms Module Testing Study Consortium Health-related quality of life in pediatric patients with functional and organic gastrointestinal diseases. J Pediatr. 2015;166(1):85–90. doi: 10.1016/j.jpeds.2014.08.022.S0022-3476(14)00748-3 - DOI - PubMed
-
- Sjölund J, Uusijärvi A, Tornkvist NT, Kull I, Bergström A, Alm J, Törnblom H, Olén O, Simrén M. Prevalence and progression of recurrent abdominal pain, from early childhood to adolescence. Clin Gastroenterol Hepatol. 2021;19(5):930–938.e8. doi: 10.1016/j.cgh.2020.04.047.S1542-3565(20)30592-9 - DOI - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

