Advances in subpopulation separation and detection of extracellular vesicles: for liquid biopsy and downstream research
- PMID: 39776815
- PMCID: PMC11700854
- DOI: 10.7150/thno.106459
Advances in subpopulation separation and detection of extracellular vesicles: for liquid biopsy and downstream research
Abstract
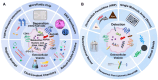
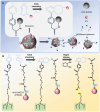
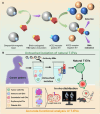
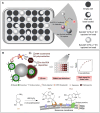
Extracellular vesicles (EVs) are carriers of a diverse array of bioactive molecules, making them valuable clinical tools for liquid biopsy in disease diagnosis and prognosis evaluation. These molecules play critical roles in various physiological and pathological conditions, and effective separation of EVs is essential to achieve these objectives. Due to the high heterogeneity of EVs, particularly with regard to their cargo molecules, merely isolating the general EV population is inadequate for liquid biopsy and biological function studies. Therefore, separating EV subpopulations becomes crucial. Traditional separation methods, such as differential ultracentrifugation and size exclusion chromatography, along with burgeoning techniques like classical microfluidic chips and covalent chemistry, often prove time-consuming, yield low purity, and have limited ability to address cargo heterogeneity. Thus, precise separation of EV subpopulations is of utmost importance. Additionally, detecting subpopulation-specific cargo is vital for validating the effectiveness of separation methods and supporting clinical biopsy applications. However, reviews that focus specifically on detection methods for EV subpopulations are limited. This paper provides a comprehensive overview of the methods for separating and detecting EV subpopulations with surface marker heterogeneity, comparing the advantages and limitations of each technique. Furthermore, it discusses challenges and future prospects for these methods in the context of liquid biopsy and downstream research. Collectively, this review aims to offer innovative insights into the separation and detection of EV subpopulations, guiding researchers to avoid common pitfalls and refine their investigative approaches.
Keywords: detection; extracellular vesicle; liquid biopsy; separation; subpopulation.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures



Similar articles
-
Separation of small extracellular vesicles (sEV) from human blood by Superose 6 size exclusion chromatography.J Extracell Vesicles. 2024 Oct;13(10):e70008. doi: 10.1002/jev2.70008. J Extracell Vesicles. 2024. PMID: 39441012 Free PMC article.
-
Extracellular Vesicle Separation Techniques Impact Results from Human Blood Samples: Considerations for Diagnostic Applications.Int J Mol Sci. 2021 Aug 26;22(17):9211. doi: 10.3390/ijms22179211. Int J Mol Sci. 2021. PMID: 34502122 Free PMC article.
-
Emerging methods in biomarker identification for extracellular vesicle-based liquid biopsy.J Extracell Vesicles. 2021 May;10(7):e12090. doi: 10.1002/jev2.12090. Epub 2021 May 12. J Extracell Vesicles. 2021. PMID: 34012517 Free PMC article. Review.
-
[Exosome separation and analysis based on microfluidics technology and its clinical applications].Se Pu. 2025 May;43(5):455-471. doi: 10.3724/SP.J.1123.2024.10032. Se Pu. 2025. PMID: 40331610 Free PMC article. Review. Chinese.
-
Extracellular vesicles: the next generation of biomarkers for liquid biopsy-based prostate cancer diagnosis.Theranostics. 2020 Jan 16;10(5):2309-2326. doi: 10.7150/thno.39486. eCollection 2020. Theranostics. 2020. PMID: 32089744 Free PMC article. Review.
Cited by
-
Salivary Extracellular Vesicles in Detection of Head and Neck Cancers: A Systematic Review.Int J Nanomedicine. 2025 May 26;20:6757-6775. doi: 10.2147/IJN.S520288. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40452788 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

