FeMOFs/CO loading reduces NETosis and macrophage inflammatory response in PLA based cardiovascular stent materials
- PMID: 39776860
- PMCID: PMC11703552
- DOI: 10.1093/rb/rbae140
FeMOFs/CO loading reduces NETosis and macrophage inflammatory response in PLA based cardiovascular stent materials
Abstract
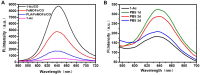
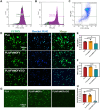
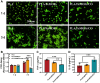
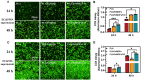
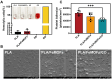
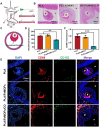
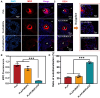
Modification of polylactic acid (PLA) is a promising strategy for the next generation of bioresorbable vascular stent biomaterials. With this focus, FeMOFs nanoparticles was incorporated in PLA, and then post loading of carbon monoxide (CO) was performed by pressurization. It showed FeMOFs incorporation increased hydrophilicity of the surface and CO loading, and CO release was sustained at least for 3 days. It is well acknowledged NETosis and macrophage mediated inflammation are the principal effectors of atherosclerosis and cardiovascular disease, and it further increases the risk of late stent thrombosis and restenosis. In this study, the effects of CO release of PLA/FeMOFs/CO on NETosis and macrophage behavior were thoroughly explored. In vitro evaluation results showed that PLA/FeMOFs/CO significantly inhibited neutrophil extracellular traps (NETs) release and neutrophil elastase expression by reducing intracellular reactive oxygen species in a simulated inflammatory environment. It reduced Lipopolysaccharide-induced macrophage inflammation with decreased tumor necrosis factor-α expression and increased IL-10 expression. Meanwhile it enhanced endothelial cell activity and growth in inflammatory environment, and inhibited platelet adhesion and activation. In vivo implantation results confirmed that PLA/FeMOFs/CO reduced the macrophages and neutrophils mediated inflammatory response, thus reduced the neointimal hyperplasia. Overall, PLA/FeMOFs/CO effectively prevented the inflammation and restenosis associated with PLA implantation. Our study provides a new strategy to improve the immunocompatibility of PLA implant materials.
Keywords: FeMOFs; NETosis; carbon monoxide; inflammation; macrophage.
© The Author(s) 2024. Published by Oxford University Press.
Figures

References
-
- Tsao CW, Aday AW, Almarzooq ZI, Alonso A, Beaton AZ, Bittencourt MS, Boehme AK, Buxton AE, Carson AP, Commodore-Mensah Y, Elkind MSV, Evenson KR, Eze-Nliam C, Ferguson JF, Generoso G, Ho JE, Kalani R, Khan SS, Kissela BM, Knutson KL, Levine DA, Lewis TT, Liu J, Loop MS, Ma J, Mussolino ME, Navaneethan SD, Perak AM, Poudel R, Rezk-Hanna M, Roth GA, Schroeder EB, Shah SH, Thacker EL, VanWagner LB, Virani SS, Voecks JH, Wang N-Y, Yaffe K, Martin SS. Heart disease and stroke statistics—2022 update: a report from the American Heart Association. Circulation 2022;145:e153–e639. - PubMed
-
- Schapiro-Dufour E, Tricotel A, Slama MS, Ducimetière P, Trinh-Duc A, Sichel C, Le Tri T, Galmiche H, Dray-Spira R, Zureik M. Major ischaemic and bleeding risks following current drug-eluting stent implantation: are there differences across current drug-eluting stent types in real life? Arch Cardiovasc Dis 2019;112:469–84. - PubMed
-
- Dworeck C, Angeras O, Haraldsson I, Ioanes D, Odenstedt J, Petursson P, Robertsson L, Stewart J, Volz S, Albertsson P, Råmunddal T, Omerovic E. TCT-477 long-term risk of stent thrombosis and restenosis after treatment with drug-eluting stents: a report from SCAAR. J Am Coll Cardiol 2016;68:B191–2.
-
- Lasprilla AJR, Martinez GAR, Lunelli BH, Jardini AL, Filho RM. Poly-lactic acid synthesis for application in biomedical devices–a review. Biotechnol Adv 2012;30:321–8. - PubMed
LinkOut - more resources
Full Text Sources

