Ionizing radiation improves skin bacterial dysbiosis in cutaneous T-cell lymphoma
- PMID: 39776918
- PMCID: PMC11703887
- DOI: 10.3389/fimmu.2024.1520214
Ionizing radiation improves skin bacterial dysbiosis in cutaneous T-cell lymphoma
Abstract
Introduction: Cutaneous T-cell lymphoma (CTCL) is closely associated with the host microbiome. While recent evidence suggests that shifts in specific bacterial taxa are associated with response to UV-B, a form of non-ionizing radiation, the impact of ionizing radiation (IR) has not been investigated.
Methods: 16S rRNA and tuf gene amplicon sequencing were performed on DNA extracted from swabs of lesional/non-lesional skin of 12 CTCL patients before/after TSEBT or local IR and from 25 matched healthy controls (HC). Microbial diversity and taxonomic profiles were analyzed.
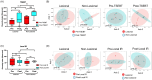
Results: Radiation exposure increased CTCL skin α-diversity to levels approximating HC. TSEBT appeared to carry the greatest effect compared to local IR. Both α and β-diversity differed significantly post versus pre-IR for TSEBT, but not for local IR. IR was associated with decreases in known pathogenic bacteria such as Streptococcus and S. aureus and increases in healthy commensal bacteria such as Anaerococcus, Bifidobacterium and commensal staphylococci including S. pettenkoferi. Substantially more taxa shifts were seen with TSEBT versus local IR.
Discussion: IR not only eliminates CTCL lesions via induction of apoptosis, but also facilitates skin barrier restoration and recolonization of bacterial taxa associated with a healthy skin microbiome. Local IR does not have as strong an effect on the skin microbiome as TSEBT. As skin microbiota act as immunomodulators with local and potentially systemic influence, TSEBT may also improve CTCL lesions via global effects on the skin microbiome. Future larger-scale studies are required to fully elucidate the relationship between cutaneous microbes and IR treatment in CTCL.
Keywords: cutaneous T-cell lymphoma; lymphoma; microbiome; radiation; radiotherapy; skin cancer; total skin electron beam therapy.
Copyright © 2024 Chrisman, Pang, Hooper, Rajeev-Kumar, Nguyen, Green, Seed, Liang, Mittal, Hasan, Guitart, Weichselbaum, Burns and Zhou.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Narrowband ultraviolet B response in cutaneous T-cell lymphoma is characterized by increased bacterial diversity and reduced Staphylococcus aureus and Staphylococcus lugdunensis.Front Immunol. 2022 Nov 11;13:1022093. doi: 10.3389/fimmu.2022.1022093. eCollection 2022. Front Immunol. 2022. PMID: 36439132 Free PMC article.
-
Gut microbiota analyses of cutaneous T-cell lymphoma patients undergoing narrowband ultraviolet B therapy reveal alterations associated with disease treatment.Front Immunol. 2024 Jan 11;14:1280205. doi: 10.3389/fimmu.2023.1280205. eCollection 2023. Front Immunol. 2024. PMID: 38274799 Free PMC article.
-
Gut dysbiosis in cutaneous T-cell lymphoma is characterized by shifts in relative abundances of specific bacterial taxa and decreased diversity in more advanced disease.J Eur Acad Dermatol Venereol. 2022 Sep;36(9):1552-1563. doi: 10.1111/jdv.18125. Epub 2022 Apr 19. J Eur Acad Dermatol Venereol. 2022. PMID: 35366365 Free PMC article.
-
Total Skin Electron Beam Therapy as Part of Multimodal Treatment Strategies for Primary Cutaneous T-Cell Lymphoma.Oncol Res Treat. 2017;40(5):244-252. doi: 10.1159/000475634. Epub 2017 Apr 25. Oncol Res Treat. 2017. PMID: 28448985 Review.
-
Breaking Down the Barriers for Patients With Cutaneous T-Cell Lymphoma: Current Controversies and Challenges for Radiation Oncologists in 2024.Semin Radiat Oncol. 2025 Jan;35(1):110-125. doi: 10.1016/j.semradonc.2024.08.005. Semin Radiat Oncol. 2025. PMID: 39672636 Review.
References
-
- Dulmage BO, Kong BY, Holzem K, Guitart J. What is new in CTCL—Pathogenesis, diagnosis, and treatments. Curr Dermatol Rep. (2018) 7:91–8. doi: 10.1007/s13671-018-0214-0 - DOI
-
- Hooper MJ, LeWitt TM, Pang Y, Veon FL, Chlipala GE, Feferman L, et al. . Gut dysbiosis in cutaneous T-cell lymphoma is characterized by shifts in relative abundances of specific bacterial taxa and decreased diversity in more advanced disease. J Eur Acad Dermatol Venereol. (2022) 36:1552–63. doi: 10.1111/jdv.18125 - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

