A fast and responsive turn-on fluorescent probe based on a quinone conjugated alkoxy derivative for biothiols and a cellular imaging study
- PMID: 39780844
- PMCID: PMC11706294
- DOI: 10.55730/1300-0527.3702
A fast and responsive turn-on fluorescent probe based on a quinone conjugated alkoxy derivative for biothiols and a cellular imaging study
Abstract
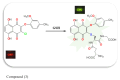
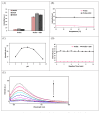
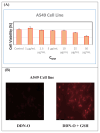
The detection of intracellular biothiols (cysteine, N-acetyl cysteine, and glutathione) with high selectivity and sensitivity is important to reveal biological functions. In this study, a 2-(2-methoxy-4-methylphenoxy)-3-chloro-5,8-dihydroxynaphthalene-1,4-dione (DDN-O) compound (3) was newly synthesized and used as a fluorogenic probe (detector molecule) in the fluorometric method for the rapid, highly selective, and sensitive determination of biothiols. The intensity values (λex = 260 nm, λem = 620 nm) of the product were measured by adding biothiols to the reaction medium at varying concentrations and the glutathione equivalent thiol content values of each biothiol were calculated. Using compound 3, glutathione as the reference biothiol was detected in the linear concentration range of 10-70 μM and the LOD value was found to be 0.11 μM. Biothiol detection with structurally simple compound 3 was performed at the cellular level within 1 min and the probe was also successfully used in bioimaging with low cytotoxicity. It was concluded that this probe can serve as an alternative to existing fluorescence-based biothiol probes with applications in rapid biothiol detection at the cellular level for biological functions. To evaluate the molecular structure of 3, conformational analysis was performed using the PM3 semiempirical method. The most stable obtained molecular geometry was then optimized at the DFT/wb97xd/6-311++G(d,p) level of theory. Frontier molecular orbitals (HOMO and LUMO) and molecular electrostatic potential map analyses were performed for the optimized structure. Molecular docking studies demonstrated the interactions of 3 with HAS (1AO6) and FhGST (2FHE) target proteins.
Keywords: 2; 3-dichloro-5; 4-dione; 8-dihydroxynaphthalene-1; Biothiols; bioimaging; fluorogenic probe; molecular docking; molecular modeling.
© TÜBİTAK.
Figures


Similar articles
-
A New Quinone Based Fluorescent Probe for High Sensitive and Selective Detection of Biothiols and Its Application in Living Cell Imaging.Int J Anal Chem. 2019 Apr 9;2019:7536431. doi: 10.1155/2019/7536431. eCollection 2019. Int J Anal Chem. 2019. PMID: 31093288 Free PMC article.
-
2,4-Dinitrobenzenesulfonate-functionalized carbon dots as a turn-on fluorescent probe for imaging of biothiols in living cells.Mikrochim Acta. 2019 Jun 10;186(7):402. doi: 10.1007/s00604-019-3503-9. Mikrochim Acta. 2019. PMID: 31183577
-
Thiol-chromene click reaction-triggered mitochondria-targeted ratiometric fluorescent probe for intracellular biothiol imaging.Anal Bioanal Chem. 2024 Nov;416(28):6223-6235. doi: 10.1007/s00216-024-05506-3. Epub 2024 Aug 30. Anal Bioanal Chem. 2024. PMID: 39212698
-
Recent Progress in the Rational Design of Biothiol-Responsive Fluorescent Probes.Molecules. 2023 May 22;28(10):4252. doi: 10.3390/molecules28104252. Molecules. 2023. PMID: 37241992 Free PMC article. Review.
-
Nucleophilicity of cysteine and related biothiols and the development of fluorogenic probes and other applications.Org Biomol Chem. 2020 Dec 7;18(46):9398-9427. doi: 10.1039/d0ob01754j. Org Biomol Chem. 2020. PMID: 33200155 Review.
References
LinkOut - more resources
Full Text Sources
