Targeting WDR5/ATAD2 signaling by the CK2/IKAROS axis demonstrates therapeutic efficacy in T-ALL
- PMID: 39785511
- PMCID: PMC11969266
- DOI: 10.1182/blood.2024024130
Targeting WDR5/ATAD2 signaling by the CK2/IKAROS axis demonstrates therapeutic efficacy in T-ALL
Abstract
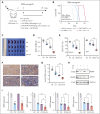
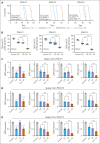
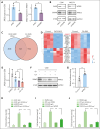
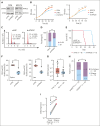
T-cell acute lymphoblastic leukemia (T-ALL) is an aggressive hematological malignancy with a poor prognosis and limited options for targeted therapies. Identifying new molecular targets to develop novel therapeutic strategies is the pressing immediate issue in T-ALL. Here, we observed high expression of WD repeat-containing protein 5 (WDR5) in T-ALL. With in vitro and in vivo models, we demonstrated the oncogenic role of WDR5 in T-ALL by activating cell cycle signaling through its new downstream effector, ATPase family AAA domain-containing 2 (ATAD2). Moreover, the function of a zinc finger transcription factor of the Kruppel family (IKAROS) is often impaired by genetic alteration and casein kinase II (CK2) which is overexpressed in T-ALL. We found that IKAROS directly regulates WDR5 transcription; CK2 inhibitor, CX-4945, strongly suppresses WDR5 expression by restoring IKAROS function. Last, combining CX-4945 with WDR5 inhibitor demonstrates synergistic efficacy in the patient-derived xenograft mouse models. In conclusion, our results demonstrated that WDR5/ATAD2 is a new oncogenic signaling pathway in T-ALL, and simultaneous targeting of WRD5 and CK2/IKAROS has synergistic antileukemic efficacy and represents a promising potential strategy for T-ALL therapy.
© 2025 American Society of Hematology. Published by Elsevier Inc. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Figures
Comment in
-
Two is better than one: dual targeting of WDR5 in T-ALL.Blood. 2025 Mar 27;145(13):1339-1341. doi: 10.1182/blood.2024027633. Blood. 2025. PMID: 40146156 No abstract available.
References
-
- Vadillo E, Dorantes-Acosta E, Pelayo R, Schnoor M. T cell acute lymphoblastic leukemia (T-ALL): new insights into the cellular origins and infiltration mechanisms common and unique among hematologic malignancies. Blood Rev. 2018;32(1):36–51. - PubMed
-
- Patel AA, Thomas J, Rojek AE, Stock W. Biology and treatment paradigms in T cell acute lymphoblastic leukemia in older adolescents and adults. Curr Treat Options Oncol. 2020;21(7):57. - PubMed
-
- Pui CH, Evans WE. Treatment of acute lymphoblastic leukemia. N Engl J Med. 2006;354(2):166–178. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

