A PAS-targeting hERG1 activator reduces arrhythmic events in Jervell and Lange-Nielsen syndrome patient-derived hiPSC-CMs
- PMID: 39786967
- PMCID: PMC11949009
- DOI: 10.1172/jci.insight.183444
A PAS-targeting hERG1 activator reduces arrhythmic events in Jervell and Lange-Nielsen syndrome patient-derived hiPSC-CMs
Abstract
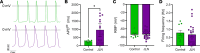
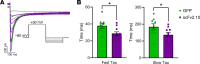
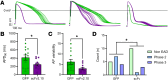
The hERG1 potassium channel conducts the cardiac repolarizing current, IKr. hERG1 has emerged as a therapeutic target for cardiac diseases marked by prolonged action potential duration (APD). Unfortunately, many hERG1 activators display off-target and proarrhythmic effects that limit their therapeutic potential. A Per-Arnt-Sim (PAS) domain in the hERG1 N-terminus reduces IKr by slowing channel activation and promoting inactivation. Disrupting PAS activity increases IKr and shortens APD in human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs). We thus hypothesized that the hERG1 PAS domain could represent a therapeutic target to reduce arrhythmogenic potential in a long QT syndrome (LQTS) background. To test this, we measured the antiarrhythmic capacity of a PAS-disabling single-chain variable fragment antibody, scFv2.10, in a hiPSC-CM line derived from a patient with Jervell and Lange Nielsen (JLN) syndrome. JLN is a severe form of LQTS caused by autosomal recessive mutations in KCNQ1. The patient in this study carried compound heterozygous mutations in KCNQ1. Corresponding JLN hiPSC-CMs displayed prolonged APD and early afterdepolarizations (EADs). Disrupting PAS with scFv2.10 increased IKr, shortened APD, and reduced the incidence of EADs. These data demonstrate that the hERG1 PAS domain could serve as a therapeutic target to treat disorders of cardiac electrical dysfunction.
Keywords: Arrhythmias; Cardiology; Ion channels; Potassium channels.
Conflict of interest statement
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

