Nanostructured lipid carriers based mRNA vaccine leads to a T cell-inflamed tumour microenvironment favourable for improving PD-1/PD-L1 blocking therapy and long-term immunity in a cold tumour model
- PMID: 39793480
- PMCID: PMC11774803
- DOI: 10.1016/j.ebiom.2024.105543
Nanostructured lipid carriers based mRNA vaccine leads to a T cell-inflamed tumour microenvironment favourable for improving PD-1/PD-L1 blocking therapy and long-term immunity in a cold tumour model
Abstract
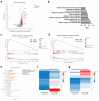
Background: mRNA-based cancer vaccines show promise in triggering antitumour immune responses. To combine them with existing immunotherapies, the intratumoral immune microenvironment needs to be deeply characterised. Here, we test nanostructured lipid carriers (NLCs), the so-called Lipidots®, for delivering unmodified mRNA encoding Ovalbumin (OVA) antigen to elicit specific antitumour responses.
Methods: We evaluated whether NLC OVA mRNA complexes activate dendritic cells (DCs) in vitro and identified the involved signalling pathways using specific inhibitors. We tested the anti-tumoral impact of Ova mRNA vaccine in B16-OVA and E.G7-OVA cold tumour-bearing C57Bl6 female mice as well as its synergy effect with an anti-PD-1 therapy by following the tumour growth and performing immunophenotyping of innate and adaptive immune cells. The intratumoral vaccine-related gene signature was assessed by RNA-sequencing. The immune memory response was assessed by re-challenging surviving treated mice with tumour cells.
Findings: Our vaccine activates DCs in vitro through the TLR4/8 and ROS signalling pathways and induces specific T cell activation while exhibits significant preventive and therapeutic antitumour efficacy in vivo. A profound intratumoral remodelling of the innate and adaptive immunity in association with an increase in the gene expression of chemokines (Cxcl10, Cxcl11, Cxcl9) involved in CD8+ T cell attraction were observed in immunised mice. The combination of vaccine and anti-PD-1 therapy improves the rates of complete responses and memory immune responses compared to monotherapies.
Interpretation: Lipidots® are effective platform for the development of vaccines against cancer based on mRNA delivery. Their combination with immune checkpoint blockers could counter tumour resistance and promote long-term antitumour immunity.
Funding: This work was supported by Inserm Transfert, la Région Auvergne Rhône Alpes, FINOVI, and the French Ministry of Higher Education, research and innovation (LipiVAC, COROL project, funding reference N° 2102992411).
Keywords: Cancer; Immune checkpoint inhibitor; Innate immunity; Memory immunity; T cells; mRNA vaccine.
Copyright © 2025 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of interests T.D. declares consulting fees (BMS, AstraZeneca, Roche, BD), honoraria for lecture and presentations (BMS, Abbvie, Gilead, AstraZeneca, Roche), payment for expert testimony (HAS) and support for attending meeting (MSD, Abbvie, Gilead, AstraZeneca, Roche). The other authors declare that they have no competing interests.
Figures





References
-
- Vinay D.S., Ryan E.P., Pawelec G., et al. Immune evasion in cancer: mechanistic basis and therapeutic strategies. Semin Cancer Biol. 2015;35(Suppl):S185–S198. - PubMed
-
- Reck M., Rodríguez-Abreu D., Robinson A.G., et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375(19):1823–1833. - PubMed
-
- Schachter J., Ribas A., Long G.V., et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006) Lancet. 2017;390(10105):1853–1862. - PubMed
-
- Rini B.I., Powles T., Atkins M.B., et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet. 2019;393(10189):2404–2415. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

