Indocarbocyanine-Indodicarbocyanine (sCy3-sCy5) Absorptive Interactions in Conjugates and DNA Duplexes
- PMID: 39795114
- PMCID: PMC11721635
- DOI: 10.3390/molecules30010057
Indocarbocyanine-Indodicarbocyanine (sCy3-sCy5) Absorptive Interactions in Conjugates and DNA Duplexes
Abstract
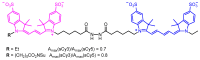
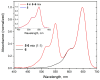
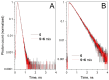
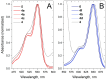
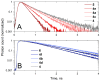
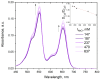
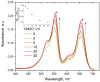
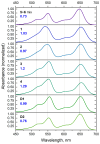
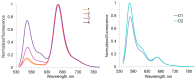
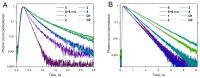
Sulfonated indocyanines 3 and 5 (sCy3, sCy5) are widely used to label biomolecules. Their high molar absorption coefficients and lack of spectral overlap with biopolymers make them ideal as linker components for rapid assessment of bioconjugate stoichiometry. We recently found that the determination of the sCy3:sCy5 molar ratio in a conjugate from its optical absorption spectrum is not straightforward, as the sCy3:sCy5 absorbance ratio at the maxima tends to be larger than expected. In this work, we have investigated this phenomenon in detail by studying the spectral properties of a series of sCy3-sCy5 conjugates in which the dyes are separated by linkers of various lengths, including DNA duplexes. It was found that when sCy3 and sCy5 are located in close proximity, they consistently exhibit an "abnormal" absorbance ratio. However, when the two dyes are separated by long rigid DNA-based spacers, the absorbance ratio becomes consistent with their individual molar absorption coefficients. This phenomenon should be taken into account when assessing the molar ratio of the dyes by UV-Vis spectroscopy.
Keywords: absorption spectra; dyes interaction; energy transfer; oligonucleotide duplex; sulfocyanine dyes.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
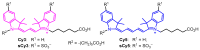

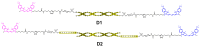
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

