Folic Acid and Methyltetrahydrofolate Supplementation in the Mthfr677C>T Mouse Model with Hepatic Steatosis
- PMID: 39796516
- PMCID: PMC11723006
- DOI: 10.3390/nu17010082
Folic Acid and Methyltetrahydrofolate Supplementation in the Mthfr677C>T Mouse Model with Hepatic Steatosis
Abstract
Background/objectives: The MTHFR677C>T gene variant results in a thermolabile MTHFR enzyme associated with elevated plasma homocysteine in TT individuals. Health risks associated with the TT genotype may be modified by dietary and supplemental folate intake. Supplementation with methyltetrahydrofolate (methylTHF) may be preferable to folic acid because it is the MTHFR product, and does not require reduction by DHFR to enter one-carbon folate metabolism. In the Mthfr677C>T mouse model for this variant, female 677TT (TT) mice have an increased incidence of hepatic steatosis. The objective of this study was to compare the effects of methylTHF and folic acid supplementation on hepatic steatosis and one-carbon metabolism in this model.
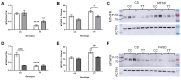
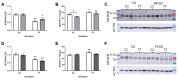
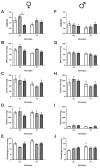
Methods: Male and female C57BL/6J 677CC (CC) and TT mice were fed control (CD), 5xmethylTHF-supplemented (MFSD), or 5xfolic-acid-supplemented (FASD) diets for 4 months. Liver sections were assessed for steatosis by Oil Red O staining. One-carbon metabolites were measured in the liver and plasma. MTHFR protein expression was evaluated in the liver.
Results: MFSD had no significant effect on plasma homocysteine, liver SAM/SAH ratios, or hepatic steatosis in males or females as compared to CD. MTHFR protein increased in MFSD TT female liver, but remained <50% of the CC. FASD had no effect on plasma homocysteine but it decreased the liver MTHFR protein and SAM/SAH ratios, and increased hepatic steatosis in CC females.
Conclusions: MethylTHF and folic acid supplementation had limited benefits for TT mice, while folic acid supplementation had negative effects on CC females. Further investigation is required to determine if these effects are relevant in humans.
Keywords: MTHFR; folic acid; hepatic steatosis; methyltetrahydrofolate; mouse model; supplementation.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
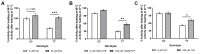

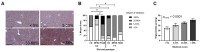
References
-
- Zhao L., Li T., Dang M., Li Y., Fan H., Hao Q., Song D., Lu J., Lu Z., Jian Y., et al. Association of methylenetetrahydrofolate reductase (MTHFR) rs1801133 (677C>T) gene polymorphism with ischemic stroke risk in different populations: An updated meta-analysis. Front. Genet. 2022;13:1021423. doi: 10.3389/fgene.2022.1021423. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

