This is a preprint.
Norditerpene natural products from subterranean fungi with anti-parasitic activity
- PMID: 39803491
- PMCID: PMC11722346
- DOI: 10.1101/2025.01.02.631097
Norditerpene natural products from subterranean fungi with anti-parasitic activity
Abstract
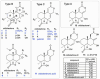
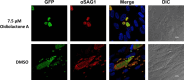
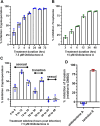
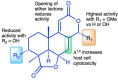
Cryptosporidium is a common, waterborne gastrointestinal parasite that causes diarrheal disease worldwide. Currently there are no effective therapeutics to treat cryptosporidiosis in at-risk populations. Since natural products are a known source of anti-parasitic compounds, we screened a library of extracts and pure natural product compounds isolated from bacteria and fungi collected from subterranean environments for activity against Cryptosporidium parvum. Eight structurally related norditerpene lactones isolated from the fungus Oidiodendron truncatum collected from the Soudan Iron mine in Minnesota showed potent activity and were further tested to identify the most active compounds. The availability of a diverse suite of natural structural analogs with varying activities allowed us to determine some structure activity relationships for both anti-parasitic activity as well as cytotoxicity. The two most potent compounds, oidiolactones A and B, had EC50s against intracellular Cryptosporidium parvum of 530 and 240 nM respectively without cytotoxicity to confluent HCT-8 host cells. Both compounds also inhibited the related parasite Toxoplasma gondii. Oidiolactone A was active against asexual, but not sexual, stages of C. parvum, and killed 80% of the parasites within 8 hours of treatment. This compound reduced C. parvum infection by 70% in IFNγ-/- mice, with no signs of toxicity. The high potency, low cytotoxicity, and in vivo activity combined with high production, easy isolation from fungi, and synthetic accessibility make oidiolactones A and B attractive scaffolds for the development of new anti-Cryptosporidium therapeutics.
Conflict of interest statement
Competing interests: The authors declare that they have no competing interests.
Figures


Similar articles
-
Novel acyl carbamates and acyl / diacyl ureas show in vitro efficacy against Toxoplasma gondii and Cryptosporidium parvum.Int J Parasitol Drugs Drug Resist. 2020 Dec;14:80-90. doi: 10.1016/j.ijpddr.2020.08.006. Epub 2020 Aug 25. Int J Parasitol Drugs Drug Resist. 2020. PMID: 33011650 Free PMC article.
-
Antifungal Norditerpene Oidiolactones from the Fungus Oidiodendron truncatum, a Potential Biocontrol Agent for White-Nose Syndrome in Bats.J Nat Prod. 2020 Feb 28;83(2):344-353. doi: 10.1021/acs.jnatprod.9b00789. Epub 2020 Jan 27. J Nat Prod. 2020. PMID: 31986046
-
Tyrosine Kinase Inhibitors Display Potent Activity against Cryptosporidium parvum.Microbiol Spectr. 2023 Feb 14;11(1):e0387422. doi: 10.1128/spectrum.03874-22. Epub 2022 Dec 19. Microbiol Spectr. 2023. PMID: 36533912 Free PMC article.
-
Apicomplexan infections in the gut.Parasite Immunol. 2014 Sep;36(9):409-20. doi: 10.1111/pim.12115. Parasite Immunol. 2014. PMID: 25201405 Review.
-
The Mucosal Innate Immune Response to Cryptosporidium parvum, a Global One Health Issue.Front Cell Infect Microbiol. 2021 May 25;11:689401. doi: 10.3389/fcimb.2021.689401. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34113580 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
