Evaluating Avacopan in the Treatment of ANCA-Associated Vasculitis: Design, Development and Positioning of Therapy
- PMID: 39803605
- PMCID: PMC11717651
- DOI: 10.2147/DDDT.S341842
Evaluating Avacopan in the Treatment of ANCA-Associated Vasculitis: Design, Development and Positioning of Therapy
Abstract
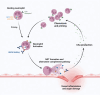
Recently, avacopan has been approved for the treatment of ANCA-associated vasculitis (AAV). Avacopan is an inhibitor of the C5a-receptor, which plays an important role in chemotaxis and the amplification loop of inflammation in AAV. In the most recent, international guidelines avacopan is recommended as steroid-sparing agents for the management of AAV. Here, we review the clinical trials that have led to demonstrate that avacopan is an effective treatment option in the management of AAV, where it can significantly reduce the cumulative dosage of glucocorticoids (GC). Despite the new guideline recommendations, clear guidance on how to employ avacopan in real-world clinical practice is lacking. We therefore also address in this review the data and clinical experience with avacopan obtained from real-world evidence. Combining preclinical studies, clinical trials, and real-world evidence helps to provide a better position of avacopan for the management of AAV in routine clinical practice, taking advantage of the GC-sparing effects of avacopan as a possible solution for the current challenge of reducing GC-toxicity in AAV patients. Furthermore, we delineate current knowledge gaps and future research areas that need to be addressed.
Keywords: ANCA; C5a inhibition; antineutrophil cytoplasmic antibody; complement; glucocorticoid toxicity; pauci-immune glomerulonephritis.
© 2025 van Leeuwen et al.
Conflict of interest statement
The work of YKOT is supported by the Arthritis Research and Collaboration Hub (ARCH) foundation. ARCH is funded by Dutch Arthritis Foundation (ReumaNederland). YKOT received an unrestricted research grant from GlaxoSmithKline, Aurinia Pharmaceuticals and Vifor Pharma. The LUMC received consulting fees from Aurinia Pharmaceuticals, Novartis, GSK, KezarBio, Vifor Pharma, Otsuka Pharmaceuticals on consultancies delivered by YKOT. LQ reports personal fees from CSL Vifor, outside the submitted work. IG reports on advisory board of CSL Vifor Pharma, during the conduct of the study. The authors report no other conflicts of interest in this work.
Figures
References
-
- Floege J, Jayne DRW, Sanders J-SF, Tesar V, Rovin BH. Kidney Disease: improving Global Outcomes AVWG. KDIGO 2024 Clinical Practice Guideline for the Management of Antineutrophil Cytoplasmic Antibody (ANCA)-Associated Vasculitis. Kidney Int. 2024;105(3S):S71–S116. doi:10.1016/j.kint.2023.10.008 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

