Impact of Fc-gamma receptor IIIA polymorphism on late-onset neutropenia and clinical outcomes in kidney transplant recipients following rituximab induction therapy
- PMID: 39804516
- PMCID: PMC12049374
- DOI: 10.1007/s10157-024-02610-7
Impact of Fc-gamma receptor IIIA polymorphism on late-onset neutropenia and clinical outcomes in kidney transplant recipients following rituximab induction therapy
Abstract
Background: This study aimed to investigate the association between the Fc-gamma receptor IIIA (FCGR3A) 158 polymorphism and clinical outcomes in kidney transplantation (KTx) patients. Specifically, we focused on late-onset neutropenia (LON) in ABO-incompatible (ABOi) or HLA-incompatible (HLAi) KTx recipients who underwent rituximab (RTx) desensitization therapy.
Methods: FCGR3A 158F/V polymorphisms were identified in 85 ABOi or HLAi KTx recipients who underwent RTx desensitization at our institution between April 2008 and October 2021. We analyzed these polymorphism groups in relation to their preoperative background and incidence of LON, infection, and rejection. In addition, we examined the risk factors for LON development.
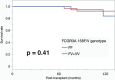
Results: The following FCGR3A 158F/V polymorphisms were identified: FF genotype (n = 45); FV genotype (n = 36), and VV genotype (n = 4). LON occurred in 25 out of 85 recipients within 1 year after KTx, significantly more frequently in patients with the FCGR3A FV + VV genotype (17/40) than in those with the FF genotype (8/45) (p = 0.01). A multivariate analysis identified the V-allele as an independent risk factor for LON (OR, 4.03; 95% CI, 1.38-11.73, p = 0.01). However, there were no significant differences in the incidence rates of post-transplant infection and rejection between the FF and FV + VV genotypes.
Conclusion: Recipients with the FCGR3A 158 V-allele were identified as having a higher risk of developing LON following KTx with RTx desensitization therapy. However, the presence of this V-allele did not affect the safety or efficacy of RTx desensitization before KTx.
Keywords: Fc-gamma receptor IIIa; Kidney transplantation; Neutropenia; Polymorphisms; Rituximab.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: The authors have declared that no conflict of interest exists. Ethical approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board (IRB) of Kobe University Hospital (IRB approval number B220142) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The studies were in accordance with the Declaration of Istanbul. Informed consent: Written informed consent was obtained from all individual participants included in the study.
Figures
Similar articles
-
Late-onset neutropenia and acute rejection in ABO-incompatible kidney transplant recipients receiving rituximab and mycophenolate mofetil.Transpl Immunol. 2014 Aug;31(2):92-7. doi: 10.1016/j.trim.2014.06.001. Epub 2014 Jun 13. Transpl Immunol. 2014. PMID: 24932811
-
Results of a multicenter prospective clinical study in Japan for evaluating efficacy and safety of desensitization protocol based on rituximab in ABO-incompatible kidney transplantation.Clin Exp Nephrol. 2017 Aug;21(4):705-713. doi: 10.1007/s10157-016-1321-5. Epub 2016 Aug 17. Clin Exp Nephrol. 2017. PMID: 27534951
-
Single fixed low-dose rituximab as induction therapy suppresses de novo donor-specific anti-HLA antibody production in ABO compatible living kidney transplant recipients.PLoS One. 2019 Oct 23;14(10):e0224203. doi: 10.1371/journal.pone.0224203. eCollection 2019. PLoS One. 2019. PMID: 31644555 Free PMC article.
-
Clinical outcomes after ABO-incompatible renal transplantation: a systematic review and meta-analysis.Lancet. 2019 May 18;393(10185):2059-2072. doi: 10.1016/S0140-6736(18)32091-9. Epub 2019 Apr 18. Lancet. 2019. PMID: 31006573
-
Anti-CD20 Blocker Rituximab in Kidney Transplantation.Transplantation. 2018 Jan;102(1):44-58. doi: 10.1097/TP.0000000000001849. Transplantation. 2018. PMID: 28614191 Review.
Cited by
-
Late-Onset Neutropenia Induced by Rituximab in Rheumatic Diseases: A Report of Two Cases of Severe Presentation and a Literature Review.Cureus. 2025 Mar 5;17(3):e80074. doi: 10.7759/cureus.80074. eCollection 2025 Mar. Cureus. 2025. PMID: 40190867 Free PMC article.
References
-
- Ko EJ, Yu JH, Yang CW, Chung BH, the Korean Organ Transplantation Registry Study Group. Clinical outcomes of ABO- and HLA-incompatible kidney transplantation: a nationwide cohort study. Transpl Int. 2017;30(12):1215–25. - PubMed
-
- Hurst FP, Belur P, Nee R, Agodoa LY, Patel P, Abbott KC, et al. Poor outcomes associated with neutropenia after kidney transplantation: Analysis of United States renal data system. Transplantation. 2011;92(1):36–40. - PubMed
-
- Koene HR, Kleijer M, Algra J, Roos D, Kr EG, Von Dem Borne A, De Haas M. FcγRIIIa-158V/F Polymorphism Influences the Binding of IgG by Natural Killer Cell FcγRIIIa Independently of the FcγRIIIa-48L/R/H Phenotype. Blood. 1997;90(3):1109–14. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous