Clinical criteria for limbic-predominant age-related TDP-43 encephalopathy
- PMID: 39807681
- PMCID: PMC11772733
- DOI: 10.1002/alz.14202
Clinical criteria for limbic-predominant age-related TDP-43 encephalopathy
Abstract
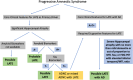
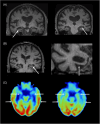
Limbic predominant age-related TDP-43 encephalopathy neuropathologic change (LATE-NC) is highly prevalent in late life and a common co-pathology with Alzheimer's disease neuropathologic change (ADNC). LATE-NC is a slowly progressive, amnestic clinical syndrome. Alternatively, when present with ADNC, LATE-NC is associated with a more rapid course. With the emergence of anti-amyloid therapeutics, discrimination of LATE-NC from ADNC is critical and will lead to greater clinical recognition of amnestic patients without ADNC. Furthermore, co-pathology with LATE-NC may influence outcomes of these therapeutics. Thus there is a need to identify patients during life with likely LATE-NC. We propose criteria for clinical diagnosis of LATE as an initial framework for further validation. In the context of progressive memory loss and substantial hippocampal atrophy, criteria are laid out for probable (amyloid negative) or possible LATE (amyloid biomarkers are unavailable or when amyloid is present, but hippocampal neurodegeneration is out of proportion to expected pure ADNC). HIGHLIGHTS: Limbic-predominant age-related TDP-43 encephalopathy (LATE) is a highly prevalent driver of neuropathologic memory loss in late life. LATE neuropathologic change (LATE-NC) is a common co-pathology with Alzheimer's disease neuropathologic change (ADNC) and may influence outcomes with emerging disease-modifying medicines. We provide initial clinical criteria for diagnosing LATE during life either when LATE-NC is the likely primary driver of symptoms or when observed in conjunction with AD. Definitions of possible and probable LATE are provided.
Keywords: Alzheimer's disease; dementia; limbic predominant age‐related TDP‐43 encephalopathy; mild cognitive impairment; multi‐etiology dementia.
© 2025 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
H.Z. has served on scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave; has given lectures in symposia sponsored by Alzecure, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, and Roche; and is a co‐founder of Brain Biomarker Solutions (BBS) in Gothenburg AB, a part of the GU Ventures Incubator Program (outside submitted work). L.G.A. has provided consultation to Eli Lilly, Biogen, Genentech, General Electric (GE) Healthcare, Eisai, Roche Diagnostics, Siemens, Alnylam, Corium, Otsuka, Two Labs, Florida Department of Health, and the National Institutes of Health (NIH) Biobank; receives research support from the Alzheimer's Association, Roche Diagnostics, Avid Pharmaceuticals, and Life Molecular Imaging; and has received honoraria for participating in independent data safety monitoring boards (DMSB) and providing educational continuing medical education (CME) lectures and programs. R.P. has served as a consultant for Roche, Genentech, Eli Lilly, Eisai, and Nestle; receives royalties from Oxford University Press and UpToDate; and has participated in educational activities with Medscape. D.A.W. has served as a paid consultant to Eli Lilly, GE Healthcare, and Qynapse; serves on a data safety monitoring board for Functional Neuromodulation and GSK; and receives research support paid to his institution from Biogen. H.‐S.Y. received honoraria from Genetech, Incorporated (Inc), unrelated to this study. D.J.I. served as unpaid member of the scientific advisory board for the Lewy Body Disorder Association (LBDA) and Denali Therapeutics. R.A.S. receives grant support from the Alzheimer's Association and GHR Foundation to her institution; grant support from Eisai and Eli Lilly to clinical trial sites; and she has served as a paid consultant for AbbVie, AC Immune, Acumen, Alector, Alnylam, Biohaven, Bristol Myers Squibb, Genentech, Jannsen, JOMDD, Nervgen, Neuroaly, Neurocentria, Oligomerix, Prothena, Renew, Roche, Shionogi, Vigil Neuroscience, Ionis, and Vaxxinity. R.M.E. is a full‐time employee of the Alzheimer's Association. Rosa R. has a patent and receives royalties from “Detecting and Treating Dementia”; has served as a consultant to Arkuda Therapeutics; is on the scientific advisory board of the Alzheimer Fondation, France and the Kissick Family Foundation Grant Program; and is on the Medical Advisory Council for the Association for Frontotemporal Degeneration. Rema R. serves as an unpaid Alzheimer's Association San Diego/Imperial Chapter Board Chair. P.S. has grant support paid to his institution from Novo Nordisk, is a member of a DSMB for ImmunoBrain Checkpoint, is Chair of the World Dementia Council, and has shares in EQT AB. He is a full‐time employee of EQT Life Sciences. E.C.M. has received consulting fees from Eli Lilly, Biogen Idec, Hoffmann‐La Roche Ltd., Janssen, and Alector. G.E.M. has served on the diversity advisory board for Genentech and the scientific advisory board for University of Texas (UT) Health Science Center at Houston; she is also on the board of Fundaconciencia Inc. and APPREMED. B.C.D. has received consulting fees from Acadia, Alector, Arkuda, Biogen, Denali, Eisai, Genentech, Lilly, Merck, Takeda, and Wave LifeSciences; royalties from Oxford University Press and Cambridge University Press; and has served on the DSMB for Eli Lilly and Merck. T.J.H. holds stock in Vivid Genomics. P.A.D. is a paid employee of the Association for Frontotemporal Degeneration. C.U.O. has grant funding paid to his institution from Alector, Inc and Transposon Therapeutics. He has served as a consultant for Acadia Pharmaceuticals, Reata Pharmaceuticals, Otsuka Pharmaceutical, Eli Lilly, Alexion Pharmaceuticals, Lykos Therapeutics, and Zevra Therapeutics. J.A.S. has served on the advisory board for the Framingham Heart Study, National Institute on Neurological Disorders and Stroke (NINDS) Discovery, and Foundation France Alzheimer's. K.D.R. received payments for presentations with MedBridge. C.M.C. serves in the DSMB for the US POnINTER study and two NIH clinical trials at the University of Kansas and University of California Los Angeles (UCLA). G.D.R. has received grant funding paid to his institution from Eli Lilly, Life Molecular Imaging, GE Healthcare, Genentch, and the Rainwater Charitable Foundation; he has received consulting fees from Eli Lilly, GE Healthcare, Roche, Genentech, and Alector; and is on a DSMB for Johnson & Johnson. R.C.P. receives grant funding to his institution from the GHR Foundation; royalties from Oxford University Press and UpToDate; and consulting fees from Roche, Genentech, Eli Lilly, Nestle, and Eisai. He is on a DSMB for Genentech and financial interests in Medscape. All other authors do not declare any conflicts of interest.
Figures
References
-
- Nelson PT, Brayne C, Flanagan ME, et al. Frequency of LATE neuropathologic change across the spectrum of Alzheimer's disease neuropathology: combined data from 13 community‐based or population‐based autopsy cohorts. Acta Neuropathol. 2022;144(1):27‐44. doi:10.1007/s00401-022-02444-1 - DOI - PMC - PubMed
Publication types
MeSH terms
Supplementary concepts
Grants and funding
- P30 AG072975/AG/NIA NIH HHS/United States
- P30 AG072946/AG/NIA NIH HHS/United States
- R01 AG080667/AG/NIA NIH HHS/United States
- R01AG034374/NH/NIH HHS/United States
- K23AG062750/NH/NIH HHS/United States
- P30 AG066530/AG/NIA NIH HHS/United States
- P01 AG066597/AG/NIA NIH HHS/United States
- R01 AG064233/AG/NIA NIH HHS/United States
- P30 AG066509/AG/NIA NIH HHS/United States
- R01 AG034374/AG/NIA NIH HHS/United States
- R01AG080667/NH/NIH HHS/United States
- RF1 AG082339/AG/NIA NIH HHS/United States
- P30AG072979/NH/NIH HHS/United States
- R01 AG072053/AG/NIA NIH HHS/United States
- K23 AG062750/AG/NIA NIH HHS/United States
- R01 AG064233/NH/NIH HHS/United States
- P01 AG066597/NH/NIH HHS/United States
- P30 AG066509/NH/NIH HHS/United States
- P30 AG072979/AG/NIA NIH HHS/United States

