The interaction of tPA with NMDAR1 drives neuroinflammation and neurodegeneration in α-synuclein-mediated neurotoxicity
- PMID: 39810216
- PMCID: PMC11731172
- DOI: 10.1186/s12974-025-03336-3
The interaction of tPA with NMDAR1 drives neuroinflammation and neurodegeneration in α-synuclein-mediated neurotoxicity
Abstract
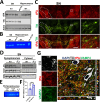
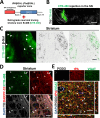
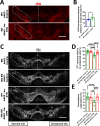
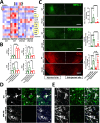
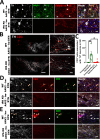
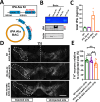
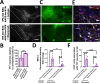
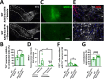
The thrombolytic protease tissue plasminogen activator (tPA) is expressed in the CNS, where it regulates diverse functions including neuronal plasticity, neuroinflammation, and blood-brain-barrier integrity. However, its role in different brain regions such as the substantia nigra (SN) is largely unexplored. In this study, we characterize tPA expression, activity, and localization in the SN using a combination of retrograde tracing and β-galactosidase tPA reporter mice. We further investigate tPA's potential role in SN pathology in an α-synuclein mouse model of Parkinson's disease (PD). To characterize the mechanism of tPA action in α-synuclein-mediated pathology in the SN and to identify possible therapeutic pathways, we performed RNA-seq analysis of the SN and used multiple transgenic mouse models. These included tPA deficient mice and two newly developed transgenic mice, a knock-in model expressing endogenous levels of proteolytically inactive tPA (tPA Ala-KI) and a second model overexpressing proteolytically inactive tPA (tPA Ala-BAC). Our findings show that striatal GABAergic neurons send tPA+ projections to dopaminergic (DA)-neurons in the SN and that tPA is released from SN-derived synaptosomes upon stimulation. We also found that tPA levels in the SN increased following α-synuclein overexpression. Importantly, tPA deficiency protects DA-neurons from degeneration, prevents behavioral deficits, and reduces microglia activation and T-cell infiltration induced by α-synuclein overexpression. RNA-seq analysis indicates that tPA in the SN is required for the upregulation of genes involved in the innate and adaptive immune responses induced by α-synuclein overexpression. Overexpression of α-synuclein in tPA Ala-KI mice, expressing only proteolytically inactive tPA, confirms that tPA-mediated neuroinflammation and neurodegeneration is independent of its proteolytic activity. Moreover, overexpression of proteolytically inactive tPA in tPA Ala-BAC mice leads to increased neuroinflammation and neurodegeneration compared to mice expressing normal levels of tPA, suggesting a tPA dose response. Finally, treatment of mice with glunomab, a neutralizing antibody that selectively blocks tPA binding to the N-methyl-D-aspartate receptor-1 (NMDAR1) without affecting NMDAR1 ion channel function, identifies the tPA interaction with NMDAR1 as necessary for tPA-mediated neuroinflammation and neurodegeneration in response to α-synuclein-mediated neurotoxicity. Thus, our data identifies a novel pathway that promotes DA-neuron degeneration and suggests a potential therapeutic intervention for PD targeting the tPA-NMDAR1 interaction.
Keywords: Dopaminergic neurons; Glunomab; Microglia; NMDAR; Neuroinflammation; Parkinson’s disease; Substantia nigra; T-cell; tPA; α-synuclein.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All animal procedures were approved by and carried out in accordance with the guidelines of the Institutional Animal Care and Use Committee at the University of Michigan. Consent for publication: Not applicable. Competing interests: Denis Vivien is a co-inventor of the patent TIE13193 (PCT#WO2014187879) concerning glunomab antibody use in neurological disorders and is President of the SAB of Lys Therapeutics. Flavie Lesept and, Nathalie Delétage are employed by Lys Therapeutics, Manuel Blanc is the Chief Executive Officer of Lys Therapeutics which is developing derivatives of Glunomab for pharmaceutical use. The remaining authors declare no competing interests.
Figures
References
-
- Baranes D, Lederfein D, Huang Y-Y, Chen M, Bailey CH, Kandel ER. Tissue plasminogen activator contributes to the late phase of LTP and to synaptic growth in the hippocampal mossy Fiber pathway. Neuron. 1998;21:813–25. - PubMed
-
- Mehra A, Ali C, Parcq J, Vivien D, Docagne F. The plasminogen activation system in neuroinflammation. Biochim et Biophys Acta (BBA) - Mol Basis Disease. 2016;1862:395–402. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

