The synergistic interaction of systemic inflammation, dysbiosis and antimicrobial resistance promotes growth restriction in children with acute severe malnutrition: An emphasis on Escherichia coli
- PMID: 39816412
- PMCID: PMC11732057
- DOI: 10.3389/frabi.2022.1001717
The synergistic interaction of systemic inflammation, dysbiosis and antimicrobial resistance promotes growth restriction in children with acute severe malnutrition: An emphasis on Escherichia coli
Abstract
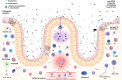
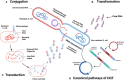
A healthy development is denied to millions of children worldwide as harsh life conditions manifest themselves in an altered inflammation-prone microbiome crosstalk environment. Keynote of this tragedy is that insufficient nutritious amino acid blocks lipids-intake to sustain diverse microbiota, and promotes the generalist strategy followed by Escherichia coli -besides other proteobacteria- of shifting gut metabolism, subverting the site specificity of first immune reaction. Furthermore, it could be hypothesized that selective success lies in their ability to induce inflammation, since this phenomenon also fuels horizontal gene transfer (HGT). In this review, we dilucidate how immune mechanisms of environmental enteric dysfunction affect overgrowth restriction, infectious morbidity rate, and acquired lifelong risks among severe acute malnourished children. Also, despite acknowledging complexities of antimicrobial resistant enrichment, we explore and speculate over the links between virulence regulation and HGT as an indissociable part in the quest for new inflammatory niches by open genome bacteria, particularly when both collide in the most vulnerable.
Keywords: Escherichia coli; antimicrobial resistance; growth restriction; gut inflammation; microbiota dysbiosis; severe acute malnutrition.
Copyright © 2022 Arredondo-Hernandez, Siebe, Castillo-Rojas, Ponce de León and López-Vidal.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The Roles of Inflammation, Nutrient Availability and the Commensal Microbiota in Enteric Pathogen Infection.Microbiol Spectr. 2015 Jun;3(3). doi: 10.1128/microbiolspec.MBP-0008-2014. Microbiol Spectr. 2015. PMID: 26185088
-
The Combined Escherichia coli Nissle 1917 and Tryptophan Treatment Modulates Immune and Metabolome Responses to Human Rotavirus Infection in a Human Infant Fecal Microbiota-Transplanted Malnourished Gnotobiotic Pig Model.mSphere. 2022 Oct 26;7(5):e0027022. doi: 10.1128/msphere.00270-22. Epub 2022 Sep 8. mSphere. 2022. PMID: 36073800 Free PMC article.
-
Dietary glycation compounds - implications for human health.Crit Rev Toxicol. 2024 Sep;54(8):485-617. doi: 10.1080/10408444.2024.2362985. Epub 2024 Aug 16. Crit Rev Toxicol. 2024. PMID: 39150724
-
The role of the gut microbiome in colonization resistance and recurrent Clostridioides difficile infection.Therap Adv Gastroenterol. 2022 Nov 18;15:17562848221134396. doi: 10.1177/17562848221134396. eCollection 2022. Therap Adv Gastroenterol. 2022. PMID: 36425405 Free PMC article. Review.
-
When a pandemic and an epidemic collide: COVID-19, gut microbiota, and the double burden of malnutrition.BMC Med. 2021 Jan 28;19(1):31. doi: 10.1186/s12916-021-01910-z. BMC Med. 2021. PMID: 33504332 Free PMC article.
Cited by
-
Bifidobacterium deficit in United States infants drives prevalent gut dysbiosis.Commun Biol. 2025 Jun 24;8(1):867. doi: 10.1038/s42003-025-08274-7. Commun Biol. 2025. PMID: 40555747 Free PMC article.
References
-
- Abdul Rahim M. B. H., Chilloux J., Martinez-Gili L., Neves A. L., Myridakis A., Gooderham N., et al. . (2019). Diet-induced metabolic changes of the human gut microbiome: Importance of short-chain fatty acids, methylamines and indoles. Acta Diabetol. 56, 493–500. doi: 10.1007/s00592-019-01312-x - DOI - PMC - PubMed
-
- Attia S., Versloot C. J., Voskuijl W., van Vliet S. J., Di Giovanni V., Zhang L., et al. . (2016). Mortality in children with complicated severe acute malnutrition is related to intestinal and systemic inflammation: An observational cohort study. Am. J. Clin. Nutr. 104, 1441–1449. doi: 10.3945/ajcn.116.130518 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

