Distinct peripheral pro-inflammatory profile associated with tuberous sclerosis complex and epilepsy
- PMID: 39817839
- PMCID: PMC11997944
- DOI: 10.1111/epi.18261
Distinct peripheral pro-inflammatory profile associated with tuberous sclerosis complex and epilepsy
Abstract
Objective: Tuberous sclerosis complex (TSC) is a monogenetic disorder associated with sustained mechanistic target of rapamycin (mTOR) activation, leading to heterogeneous clinical manifestations. Epilepsy and renal angiomyolipoma are the most important causes of morbidity in adult people with TSC (pwTSC). mTOR is a key player in inflammation, which in turn could influence TSC-related clinical manifestations. Reliable biomarkers are lacking to monitor and predict evolution and response to treatment for epilepsy in pwTSC. Inflammation has been implicated in epileptogenesis in non-TSC-related epilepsy. We aimed to characterize the relation between markers of neuroglial activation/injury, markers of peripheral inflammation, and active epilepsy in pwTSC to identify accessible biomarkers and potential new therapeutic targets.
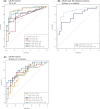
Methods: We performed a cross-sectional study to investigate markers of central nervous system (CNS) (neurofilament light [NfL] and glial fibrillary acidic protein [GFAP]) and peripheral (45 cytokines) inflammation in the peripheral blood of pwTSC (n = 46) vs age- and sex-matched healthy controls (HCs) (n = 26). In pwTSC, markers associated with active epilepsy (n = 23/46) were compared to non-TSC epilepsy controls (n = 18). Observations on markers of neuroglial activation/injury (GFAP, NfL) were confirmed in an independent TSC cohort (n = 45; 69% with active epilepsy).
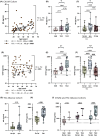
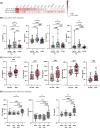
Results: We report that TSC is characterized by elevated serum levels of marker of astrogliosis (GFAP), pro-inflammatory molecules (interleukin 1β [IL-1β], CXCL8) and trophic factor (epidermal growth factor [EGF]) compared to HCs and to non-TSC-related epilepsy controls. Among pwTSC, renal angiomyolipoma presence and size was associated with IL-15. It is notable that active epilepsy in pwTSC was associated with higher levels of GFAP compared to pwTSC without epilepsy, which was confirmed in an external validation cohort, and with elevated levels of pro-inflammatory cytokines (IL-17A, IL-17C, tumor necrosis factor α [TNF-α]), not significantly related to seizure activity or treatment with mTOR inhibitor. These associations remained significant after adjusting for age and sex.
Significance: These results suggest that key inflammatory mediators could contribute to epileptogenesis and represent novel biomarkers and therapeutic targets in TSC.
Keywords: Epilepsy; GFAP; Neuroinflammation; Tuberous sclerosis complex.
© 2025 The Author(s). Epilepsia published by Wiley Periodicals LLC on behalf of International League Against Epilepsy.
Conflict of interest statement
P.M. is a local principal investigator for Zogenix (1501, 1503, 1504) and a local principal investigator for Marinus. He served as a consultant for Jazz Pharma and an invited speaker for UCB. M.R.K reports unrestricted educational grants from UCB, Eisai, Jazz Pharmaceuticals, and Paladin, as well as research grants for investigator‐initiated studies from UCB and Eisai. The remaining authors have no conflicts of interest. We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Figures


References
-
- Henske EP, Jozwiak S, Kingswood JC, Sampson JR, Thiele EA. Tuberous sclerosis complex. Nat Rev Dis Primers. 2016. May 26;2:16035. - PubMed
-
- Northrup H, Aronow ME, Bebin EM, Bissler J, Darling TN, de Vries PJ, et al. Updated international tuberous sclerosis complex diagnostic criteria and surveillance and management recommendations. Pediatr Neurol. 2021. Oct;123:50–66. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

