Reconstitution of synaptic junctions orchestrated by teneurin-latrophilin complexes
- PMID: 39818903
- PMCID: PMC11808628
- DOI: 10.1126/science.adq3586
Reconstitution of synaptic junctions orchestrated by teneurin-latrophilin complexes
Abstract
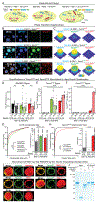
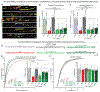
Synapses are organized by trans-synaptic adhesion molecules that coordinate assembly of pre- and postsynaptic specializations, which, in turn, are composed of scaffolding proteins forming liquid-liquid phase-separated condensates. Presynaptic teneurins mediate excitatory synapse organization by binding to postsynaptic latrophilins; however, the mechanism of action of teneurins, driven by extracellular domains evolutionarily derived from bacterial toxins, remains unclear. In this work, we show that only the intracellular sequence, a dimerization sequence, and extracellular bacterial toxin-derived latrophilin-binding domains of Teneurin-3 are required for synapse organization, suggesting that teneurin-induced latrophilin clustering mediates synaptogenesis. Intracellular Teneurin-3 sequences capture liquid-liquid phase-separated presynaptic active zone scaffolds, enabling us to reconstitute an entire synaptic junction from purified proteins in which trans-synaptic teneurin-latrophilin complexes recruit phase-separated pre- and postsynaptic specializations.
Conflict of interest statement
Figures



Similar articles
-
Latrophilins function as heterophilic cell-adhesion molecules by binding to teneurins: regulation by alternative splicing.J Biol Chem. 2014 Jan 3;289(1):387-402. doi: 10.1074/jbc.M113.504779. Epub 2013 Nov 22. J Biol Chem. 2014. PMID: 24273166 Free PMC article.
-
Signaling by latrophilin adhesion-GPCRs in synapse assembly.Neuroscience. 2025 May 24;575:150-161. doi: 10.1016/j.neuroscience.2025.03.041. Epub 2025 Mar 22. Neuroscience. 2025. PMID: 40127755 Review.
-
Latrophilin GPCRs direct synapse specificity by coincident binding of FLRTs and teneurins.Science. 2019 Feb 22;363(6429):eaav7969. doi: 10.1126/science.aav7969. Science. 2019. PMID: 30792275 Free PMC article.
-
Structural Basis of Teneurin-Latrophilin Interaction in Repulsive Guidance of Migrating Neurons.Cell. 2020 Jan 23;180(2):323-339.e19. doi: 10.1016/j.cell.2019.12.014. Epub 2020 Jan 9. Cell. 2020. PMID: 31928845 Free PMC article.
-
Teneurin Structure: Splice Variants of a Bacterial Toxin Homolog Specifies Synaptic Connections.Front Neurosci. 2019 Aug 7;13:838. doi: 10.3389/fnins.2019.00838. eCollection 2019. Front Neurosci. 2019. PMID: 31440135 Free PMC article. Review.
Cited by
-
RIM Mutants Occlude the Effects of Aliphatic Alcohols on C. elegans Locomotion.MicroPubl Biol. 2025 Jun 12;2025:10.17912/micropub.biology.001611. doi: 10.17912/micropub.biology.001611. eCollection 2025. MicroPubl Biol. 2025. PMID: 40611999 Free PMC article.
-
Cytoplasmic tail composition modulates the G protein and arrestin-3 signaling bias of the adhesion GPCR LPHN2.bioRxiv [Preprint]. 2025 Jul 6:2025.07.04.663222. doi: 10.1101/2025.07.04.663222. bioRxiv. 2025. PMID: 40631154 Free PMC article. Preprint.
-
Altered striatal dopamine regulation in ADGRL3 knockout mice.bioRxiv [Preprint]. 2025 Aug 1:2025.07.31.667389. doi: 10.1101/2025.07.31.667389. bioRxiv. 2025. PMID: 40766670 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

