Tunning valence state of cobalt centers in Cu/Co-CoO1-x for significantly boosting water-gas shift reaction
- PMID: 39820052
- PMCID: PMC11739592
- DOI: 10.1038/s41467-025-56161-w
Tunning valence state of cobalt centers in Cu/Co-CoO1-x for significantly boosting water-gas shift reaction
Abstract
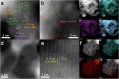
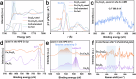
Dual active sites with synergistic valence state regulation under oxidizing and reducing conditions are essential for catalytic reactions with step-wise mechanisms to modulate the complex adsorption sites of reactant molecules on the surfaces of heterogeneous catalysts with maximized catalytic performances, but it has been rarely explored. In this work, uniformly dispersed CuCo alloy and CoO nanosheet composite catalysts with dual active sites are constructed, which shows huge boost in activity for catalyzing water-gas shift reaction (WGSR), with a record high reaction rate reaching 204.2 μmolCO gcat.-1 s-1 at 300 °C for Cu1Co9Ox amongst the reported Cu-based and Co-based catalysts. A synergistic mechanism is proposed that Coδ+ species can be easily reduced by CO adsorbed on Cu and Co0 can be oxidized by H2O. Systematic in situ characterization results reveal that the addition of Cu can regulate the redox properties of Co species and thus modulate the adsorption properties of catalysts. Particularly, doping of Cu0 sites weakens the affinity of the surface to CO or CO2 to a moderate level. Moreover, it also promotes the oxidation of *CO to *COOH and the desorption of the product CO2, reducing the carbon poisoning of the catalyst and thus increasing the reactivity. The results would provide guidance for the construction of novel heterogeneous catalyst with dual active sites and clarify its underlying reactivity enhancement mechanism induced by the tunning of valence state of metal centers for heterogeneous catalytic reactions.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Boosting reactivity of water-gas shift reaction by synergistic function over CeO2-x/CoO1-x/Co dual interfacial structures.Nat Commun. 2023 Oct 27;14(1):6851. doi: 10.1038/s41467-023-42577-9. Nat Commun. 2023. PMID: 37891176 Free PMC article.
-
Tuning the properties of copper-based catalysts based on molecular in situ studies of model systems.Acc Chem Res. 2015 Jul 21;48(7):2151-8. doi: 10.1021/acs.accounts.5b00200. Epub 2015 Jun 23. Acc Chem Res. 2015. PMID: 26103058
-
Efficient Role of Nanosheet-Like Pr2O3 Induced Surface-Interface Synergistic Structures over Cu-Based Catalysts for Enhanced Methanol Production from CO2 Hydrogenation.ACS Appl Mater Interfaces. 2022 Jan 19;14(2):2768-2781. doi: 10.1021/acsami.1c20056. Epub 2022 Jan 7. ACS Appl Mater Interfaces. 2022. PMID: 34994552
-
Insight on Atomically Dispersed Cu Catalysts for Electrochemical CO2 Reduction.ACS Nano. 2023 Oct 10;17(19):18688-18705. doi: 10.1021/acsnano.3c07307. Epub 2023 Sep 19. ACS Nano. 2023. PMID: 37725796 Review.
-
Isolated Metal Centers Activate Small Molecule Electrooxidation: Mechanisms and Applications.Adv Mater. 2025 Apr;37(15):e2418504. doi: 10.1002/adma.202418504. Epub 2025 Jan 26. Adv Mater. 2025. PMID: 39865965 Review.
References
-
- Newsome, D. S. The Water-Gas Shift Reaction. Cat. Rev.21, 275–318 (1980).
-
- Baraj, E., Ciahotny, K. & Hlincik, T. The water gas shift reaction: Catalysts and reaction mechanism. Fuel288, 119871–119886 (2021).
-
- Schlapbach, L. & Züttel, A. Hydrogen-storage materials for mobile applications. Nature414, 353–358 (2001). - PubMed
-
- Yan, H. et al. Promoted Cu-Fe3O4 catalysts for low-temperature water gas shift reaction: Optimization of Cu content. Appl. Catal. B226, 182–193 (2018).
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

