Bioleaching of lanthanum from nickel metal hydride dry battery using siderophores produced by Pseudomonas sp
- PMID: 39821467
- PMCID: PMC11739246
- DOI: 10.1007/s11274-025-04250-9
Bioleaching of lanthanum from nickel metal hydride dry battery using siderophores produced by Pseudomonas sp
Abstract
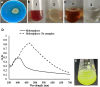
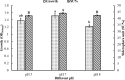
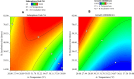
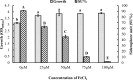
There is still much to be learned about the properties of siderophores and their applications. This study was designed to characterize and optimize the production of the siderophore produced by a marine bacterium Pseudomonas sp. strain ASA235 and then evaluate their use in bioleaching of rare earth elements (REEs) from spent Nickel-metal hydride (NiMH) batteries. The results of both Tetrazolium and Arnow's tests indicated that the test organism produces a mixed-type siderophore of pyoverdine family, a result that was confirmed by FT-IR and MALDI-TOFF analyses. Optimization of pH, temperature, incubation period, and iron concentration for siderophore production led to a noticeable shift from 44.5% up to 91% siderophore unit when the test bacterium was incubated at 28 °C and pH 7 after 72 h in the absence of iron. The purified siderophore showed the ability to bleach about 14.8% of lanthanum from the anode of the NiMH battery along with other elements, although in lower amounts. This data put siderophores in distinct focus for further prospective studies intending the bioleaching of such precious elements. The scaling up of this process and optimization would make a big difference in such a green bioleaching strategy, allowing us to recover such precious elements in an environmentally friendly way.
Keywords: Pseudomonas; Bioleaching; Lanthanum; Nickel metal hydride battery; Optimization; Siderophores..
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures

Similar articles
-
Characterization of Aspergillus niger siderophore that mediates bioleaching of rare earth elements from phosphorites.World J Microbiol Biotechnol. 2019 Jun 11;35(6):93. doi: 10.1007/s11274-019-2666-1. World J Microbiol Biotechnol. 2019. PMID: 31187335
-
Characterization of pyoverdine and achromobactin in Pseudomonas syringae pv. phaseolicola 1448a.BMC Microbiol. 2011 Oct 3;11:218. doi: 10.1186/1471-2180-11-218. BMC Microbiol. 2011. PMID: 21967163 Free PMC article.
-
Chemistry and biology of pyoverdines, Pseudomonas primary siderophores.Curr Med Chem. 2015;22(2):165-86. doi: 10.2174/0929867321666141011194624. Curr Med Chem. 2015. PMID: 25312210
-
Siderophores in fluorescent pseudomonads: new tricks from an old dog.Future Microbiol. 2007 Aug;2(4):387-95. doi: 10.2217/17460913.2.4.387. Future Microbiol. 2007. PMID: 17683275 Review.
-
Novel Insights on Pyoverdine: From Biosynthesis to Biotechnological Application.Int J Mol Sci. 2022 Sep 29;23(19):11507. doi: 10.3390/ijms231911507. Int J Mol Sci. 2022. PMID: 36232800 Free PMC article. Review.
References
-
- Ams DA, Maurice PA, Hersman LE, Forsythe JH (2002) Siderophore production by an aerobic Pseudomonas mendocina bacterium in the presence of kaolinite. Chem Geol 188:161–170. 10.1016/S0009-2541(02)00077-3
-
- Andres Y, MacCordick HJ, Hubert J-C (1991) Complexes of mycobactin from Mycobacterium smegmatis with scandium, yttrium and lanthanum. Biol Met 4:207–210. 10.1007/BF01141182 - PubMed
-
- Arnow LE (1937) Colorimetric determination of the components of 3, 4-dihydroxyphenylalanine-tyrosine mixtures. J Biol Chem 118:531–537
-
- Balk J, Schaedler TA (2014) Iron cofactor assembly in plants. Annu Rev Plant Biol 65:125–153. 10.1146/annurev-arplant-050213-035759 - PubMed
-
- Benzal E, Solé M, Lao C, Gamisans X, Dorado A (2020) Elemental copper recovery from e-wastes mediated with a two-step bioleaching process. JWBM 11:5457–5465. 10.1007/s12649-020-01040-2
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

