Toward alpha-synuclein seed amplification assay in clinical practice
- PMID: 39822296
- PMCID: PMC11736632
- DOI: 10.1002/dad2.70066
Toward alpha-synuclein seed amplification assay in clinical practice
Abstract
Introduction: Seed amplification assays (SAAs) demonstrate remarkable diagnostic performance in alpha-synucleinopathies. However, existing protocols lack accessibility in routine laboratories, mainly due to the requirement for in-house production of recombinant alpha-synuclein (aSyn). This study proposes a cerebrospinal fluid (CSF) aSyn-SAA protocol using solely commercial reagents to facilitate its clinical implementation.
Methods: Routine clinical care CSF samples from 126 patients, comprising 47 with Lewy body diseases (LBD) (41 with dementia with Lewy bodies, six with Parkinson's disease), 37 without alpha-synucleinopathy, and 42 with Alzheimer's disease (AD), underwent assessment for aSyn-SAA activity.
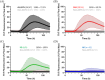
Results: CSF aSyn-SAA showed a sensitivity of 72.3% and a specificity of 100% when distinguishing clinically diagnosed LBD patients from those without alpha-synucleinopathy. In AD patients, 14.3% were tested positive for aSyn.
Discussion: The commercial-only CSF aSyn-SAA protocol exhibited excellent specificity when applied to a real-life cohort, signaling progress toward the accessibility of an aSyn biomarker in clinical settings.
Highlights: Diagnosis of LBD through aSyn-SAA lacks accessibility.This commercial-only aSyn-SAA has satisfactory performance in a real-life cohort.A negative aSyn-SAA does not completely exclude a synucleinopathy.Some technical points must be considered when developing aSyn-SAA.aSyn-SAA must be confined to expert laboratories due to prion-like risk management.
Keywords: Lewy body disease (LBD); Parkinson's disease (PD); aSyn; alpha‐synuclein (α‐synuclein); biomarker; brain homogenate (BH); cerebrospinal fluid (CSF); dementia with Lewy bodies (DLB); neurodegenerative diseases; proteinopathies; real‐life cohort; real‐time quaking‐induced conversion (RT‐QuIC); seed amplification assay (SAA); synucleinopathies.
© 2025 The Author(s). Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring published by Wiley Periodicals, LLC on behalf of Alzheimer's Association.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest. Author disclosures are available in the Supporting Information.
Figures


References
-
- Kovacs GG, Milenkovic I, Wohrer A, et al. Non‐Alzheimer neurodegenerative pathologies and their combinations are more frequent than commonly believed in the elderly brain: a community‐based autopsy series. Acta Neuropathol. 2013;126:365‐384. - PubMed
-
- Blennow K, Zetterberg H. Cerebrospinal fluid biomarkers for Alzheimer's disease. JAD. 2009;18:413‐417. - PubMed
LinkOut - more resources
Full Text Sources
