Bioinoculant-induced plant resistance is modulated by interactions with resident soil microbes
- PMID: 39827166
- PMCID: PMC11748581
- DOI: 10.1186/s40793-025-00667-9
Bioinoculant-induced plant resistance is modulated by interactions with resident soil microbes
Abstract
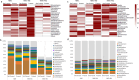
Background: Entomopathogenic fungi are increasingly used as bio-inoculants to enhance crop growth and resistance. When applied to rhizosphere soil, they interact with resident soil microbes, which can affect their ability to colonize and induce resistance in plants as well as modify the structure of the resident soil microbiome, either directly through interactions in the rhizosphere or indirectly, mediated by the plant. The extent to which such direct versus indirect interactions between bio-inoculants and soil microbes impact microbe-induced resistance in crops remains unclear. This study uses a split-root system to examine the effects of direct versus indirect (plant-mediated) interactions between an entomopathogenic fungus, Metarhizium brunneum, and resident soil microbes on induced resistance in tomato against two-spotted spider mites. Additionally, the study explores how these interactions influence the composition and diversity of soil fungal and bacterial communities.
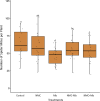
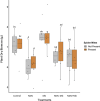
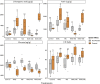
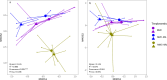
Results: Resident soil microbes reduced the efficacy of M. brunneum to induce resistance against spider mites. This reduction occurred not only when resident microbes directly interacted with the bio-inoculant but also when they were spatially separated within the root system, indicating plant-mediated effects. M. brunneum inoculation did not affect rhizosphere microbial diversity but led to changes in fungal and bacterial community composition, even when these communities were not in direct contact with the inoculant.
Conclusions: This research highlights the impact of both direct and plant-mediated interactions between bio-inoculants and resident soil microbes on bio-inoculant-induced pest resistance in crop plants and underscores the importance of assessing potential adverse effects of fungal bio-inoculants on native soil communities.
Keywords: Arthropod pests; Entomopathogenic fungi; Soil microbial communities; Spider mites; Tomato.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare that they have no competing interests.
Figures


References
-
- Meyling NV, Eilenberg J. Ecology of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae in temperate agroecosystems: potential for conservation biological control. Biol Control. 2007;43:145–55. 10.1016/j.biocontrol.2007.07.007. - DOI
-
- Jaber LR, Enkerli J. Fungal entomopathogens as endophytes: Can they promote plant growth? Biocontrol Sci Technol. 2017;27:28–41. 10.1080/09583157.2016.1243227. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
