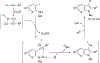
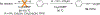One-Pot Synthesis of 2-Substituted Indoles and 7-Azaindoles via Sequential Alkynylation and Cyclization of 2-Iodo- N-mesylarylamines and Alkynes in the Presence of Cu2O
- PMID: 39830603
- PMCID: PMC11741182
- DOI: 10.1002/ajoc.202400421
One-Pot Synthesis of 2-Substituted Indoles and 7-Azaindoles via Sequential Alkynylation and Cyclization of 2-Iodo- N-mesylarylamines and Alkynes in the Presence of Cu2O
Abstract
A one-pot process was developed to synthesize in moderate to high yield a series of 2-substituted indoles and 7-azaindoles starting from 2-iodo-N-mesylarylamines and terminal alkynes in the presence of Cu2O in DMF at 90-120 °C. Without isolation of any intermediate, our optimized conditions enabled the introduction of ester, phenyl, hydroxymethyl, hydroxyethyl, N-Boc-aminomethyl, and methyl at the 2-postion of indoles and 7-azaindoles. The reaction tolerates a variety of substrates containing halogens, or acid- or base-sensitive functional groups without requiring a Pd catalyst, a ligand, or a base.
Keywords: 5-endo-dig; 7-azaindoles; Castro-Stephens; alkynes; indoles.
Figures
Similar articles
-
Gold-catalyzed sequential alkyne activation: one-pot synthesis of NH-carbazoles via cascade hydroarylation of alkyne/6-endo-dig carbocyclization reactions.J Org Chem. 2013 Jul 5;78(13):6769-74. doi: 10.1021/jo400799b. Epub 2013 Jun 21. J Org Chem. 2013. PMID: 23789909
-
Palladium-catalyzed synthesis of 2,3-disubstituted 5-azaindoles via heteroannulation reaction and of 2-substituted 5-azaindoles through domino sila-Sonogashira/5-endo cyclization.J Org Chem. 2012 Jun 1;77(11):5006-16. doi: 10.1021/jo300481s. Epub 2012 May 18. J Org Chem. 2012. PMID: 22587741
-
Efficient access to azaindoles and indoles.Org Lett. 2006 Jul 20;8(15):3307-10. doi: 10.1021/ol061232r. Org Lett. 2006. PMID: 16836392
-
Palladium complexes with an annellated mesoionic carbene (MIC) ligand: catalytic sequential Sonogashira coupling/cyclization reaction for one-pot synthesis of benzofuran, indole, isocoumarin and isoquinolone derivatives.Dalton Trans. 2020 Nov 10;49(43):15238-15248. doi: 10.1039/d0dt02918a. Dalton Trans. 2020. PMID: 33169742
-
Indole synthesis by radical cyclization of o-alkenylphenyl isocyanides and its application to the total synthesis of natural products.Chem Rec. 2002;2(1):37-45. doi: 10.1002/tcr.10008. Chem Rec. 2002. PMID: 11933260 Review.
References
-
- Saxton JE, Nat. Prod. Rep 1995, 12, 385–411; - PubMed
- Saxton JE, Nat. Prod. Rep 1994, 11, 493–531; - PubMed
- Chadha N, Silakari O, Eur. J. Med. Chem 2017, 134, 159–184; - PubMed
- Van Order RB, Lindwall HG, Chem. Rev 2002, 30, 69–96;
- Ma YM, Liang XA, Kong Y, Jia B, Agric J. Food Chem. 2016, 64, 6659–6671; - PubMed
- Kaushik NK, Kaushik N, Attri P, Kumar N, Kim CH, Verma AK, Choi EH, Molecules 2013, 18, 6620–6662; - PMC - PubMed
- Homer JA, Sperry J, J. Nat. Prod 2017, 80, 2178–2187; - PubMed
- Kumari A, Singh RK, Bioorg. Chem 2019, 89, 103021. - PubMed
-
- Chen D, Chen Y, Ma Z, Zou L, Li J, Liu Y, J. Org. Chem 2018, 83, 6805–6814; - PubMed
- Chen X, Qiu G, Liu R, Chen D, Chen Z, Org. Chem. Front 2020, 7, 890–895;
- Ganesan B, Senadi GC, Guo B-C, Hung M-Y, Lin W-Y, RSC Adv. 2018, 8, 40968–40973; - PMC - PubMed
- Ling F, Song D, Chen L, Liu T, Yu M, Ma Y, Xiao L, Xu M, Zhong W, J. Org. Chem 2020, 85, 3224–3233; - PubMed
- Peng H, Jiang K, Zhen G, Wang F, Yin B, RSC Adv. 2020, 10, 11750–11754; - PMC - PubMed
- Yuan S, Zhang DQ, Zhang JY, Yu B, Liu HM, Org. Lett 2020, 22, 814–817; - PubMed
- Zhang AA, Meng T, Wang W, Liu X, Zhu Y, Liu L, Appl. Organomet.Chem 2020, 34, e5513;
- Zhang BS, Li Y, Zhang Z, An Y, Wen YH, Gou XY, Quan SQ, Wang XG, Liang YM, J. Am. Chem. Soc 2019, 141, 9731–9738; - PubMed
- Zhao G, Roudaut C, Gandon V, Alami M, Provot O, Green Chem. 2019, 21, 4204–4210.
-
- Cacchi S, Fabrizi G, Chem. Rev 2005, 105, 2873–2920; - PubMed
- Humphrey GR, Kuethe JT, Chem. Rev 2006, 106, 2875–2911; - PubMed
- Newman SG, Lautens M, J. Am. Chem. Soc 2010, 132, 11416–11417; - PubMed
- Bugaenko DI, Karchava AV, Yurovskaya MA, Russ. Chem. Rev 2019, 88, 99–159;
- Yu Y, Zhong JS, Xu K, Yuan Y, Ye KY, Adv. Synth. Catal 2020;
- Neto JSS, Zeni G, Org. Chem. Front 2020, 7, 155–210.
-
- Larock RC, Yum EK, J. Am. Chem. Soc 1991, 113, 6689–6690.
-
- Tao SW, Zhou JY, Liu RQ, Zhu YM, J. Org. Chem 2019, 84, 8121–8130; - PubMed
- Politanskaya L, Petyuk M, Tretyakov E, J. Fluor. Chem 2019, 228.
Grants and funding
LinkOut - more resources
Full Text Sources