The interplay between mitochondrial DNA genotypes, female infertility, ovarian response, and mutagenesis in oocytes
- PMID: 39830711
- PMCID: PMC11739621
- DOI: 10.1093/hropen/hoae074
The interplay between mitochondrial DNA genotypes, female infertility, ovarian response, and mutagenesis in oocytes
Erratum in
-
Correction to: The interplay between mitochondrial DNA genotypes, female infertility, ovarian response, and mutagenesis in oocytes.Hum Reprod Open. 2025 Jul 7;2025(3):hoaf037. doi: 10.1093/hropen/hoaf037. eCollection 2025. Hum Reprod Open. 2025. PMID: 40625569 Free PMC article.
Abstract
Study question: Is there an association between different mitochondrial DNA (mtDNA) genotypes and female infertility or ovarian response, and is the appearance of variants in the oocytes favored by medically assisted reproduction (MAR) techniques?
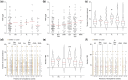
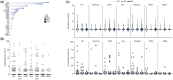
Summary answer: Ovarian response was negatively associated with global non-synonymous protein-coding homoplasmic variants but positively associated with haplogroup K; the number of oocytes retrieved in a cycle correlates with the number of heteroplasmic variants in the oocytes, principally with variants located in the hypervariable (HV) region and rRNA loci, as well as non-synonymous protein-coding variants.
What is known already: Several genes have been shown to be positively associated with infertility, and there is growing concern that MAR may facilitate the transmission of these harmful variants to offspring, thereby passing on infertility. The potential role of mtDNA variants in these two perspectives remains poorly understood.
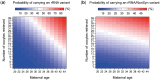
Study design size duration: This cohort study included 261 oocytes from 132 women (mean age: 32 ± 4 years) undergoing ovarian stimulation between 2019 and 2020 at an academic center. The oocyte mtDNA genotypes were examined for associations with the women's fertility characteristics.
Participants/materials setting methods: The mtDNA of the oocytes underwent deep sequencing, and the mtDNA genotypes were compared between infertile and fertile groups using Fisher's exact test. The impact of the mtDNA genotype on anti-Müllerian hormone (AMH) levels and the number of (mature) oocytes retrieved was assessed using the Mann-Whitney U test for univariate analysis and logistic regression for multivariate analysis. Additionally, we examined the associations of oocyte maturation stage, infertility status, number of ovarian stimulation units, and number of oocytes retrieved with the type and load of heteroplasmic variants using univariate analysis and Poisson or linear regression analysis.
Main results and the role of chance: Neither homoplasmic mtDNA variants nor haplogroups in the oocytes were associated with infertility status or with AMH levels. Conversely, when the relationship between the number of oocytes retrieved and different mtDNA genotypes was examined, a positive association was observed between the number of metaphase (MII) oocytes (P = 0.005) and haplogroup K. Furthermore, the presence of global non-synonymous homoplasmic variants in the protein-coding region was significantly associated with a reduced number of total oocytes and MII oocytes retrieved (P < 0.001 for both). Regarding the type and load of heteroplasmic variants in the different regions, there were no significant associations according to maturation stage of the oocyte or to fertility status; however, the number of oocytes retrieved correlated positively with the total number of heteroplasmic variants, and specifically with non-synonymous protein-coding, HV and rRNA variants (P < 0.001 for all).
Limitations reasons for caution: The current work is constrained by its retrospective design and single-center approach, potentially limiting the generalizability of our findings. The small sample size for specific types of infertility restricts this aspect of the findings.
Wider implications of the findings: This work suggests that mitochondrial genetics may have an impact on ovarian response and corroborates previous findings indicating that the size of the oocyte cohort after stimulation correlates with the presence of potentially deleterious variants in the oocyte. Future epidemiological and functional studies based on the results of the current study will provide valuable insights to address gaps in knowledge to assess any prospective risks for MAR-conceived offspring.
Study funding/competing interests: This work was supported by the Research Foundation Flanders (FWO, Grant numbers 1506617N and 1506717N to C.S.), by the Fonds Wetenschappelijk Fonds, Willy Gepts Research Foundation of Universitair Ziekenhuis Brussel (Grant numbers WFWG14-15, WFWG16-43, and WFWG19-19 to C.S.), and by the Methusalem Grant of the Vrije Universiteit Brussel (to K.S.). M.R. and E.C.d.D. were supported predoctoral fellowships by the FWO, Grant numbers 1133622N and 1S73521N, respectively. The authors declare no conflict of interests.
Trial registration number: N/A.
Keywords: haplogroup; heteroplasmy; homoplasmy; infertility; medically assisted reproduction (MAR); mitochondrial DNA; oocyte; ovarian stimulation.
© The Author(s) 2024. Published by Oxford University Press on behalf of European Society of Human Reproduction and Embryology.
Conflict of interest statement
All authors declare no conflict of interest.
Figures


Similar articles
-
Rescue in vitro maturation of germinal vesicle oocytes after ovarian stimulation: the importance of the culture media.Hum Reprod. 2025 Aug 1;40(8):1504-1515. doi: 10.1093/humrep/deaf099. Hum Reprod. 2025. PMID: 40447125 Free PMC article.
-
Pathogenic variants in DLGAP5 cause female infertility characterized by oocyte maturation arrest and embryonic arrest.Hum Reprod. 2025 Jul 10:deaf139. doi: 10.1093/humrep/deaf139. Online ahead of print. Hum Reprod. 2025. PMID: 40639803
-
Randomized controlled trial to evaluate the impact of follicle priming on IVM outcomes in women with polycystic ovaries: CFA versus FSH-B.Hum Reprod. 2025 Jun 1;40(6):1127-1137. doi: 10.1093/humrep/deaf053. Hum Reprod. 2025. PMID: 40194780 Clinical Trial.
-
Individualised gonadotropin dose selection using markers of ovarian reserve for women undergoing in vitro fertilisation plus intracytoplasmic sperm injection (IVF/ICSI).Cochrane Database Syst Rev. 2018 Feb 1;2(2):CD012693. doi: 10.1002/14651858.CD012693.pub2. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2024 Jan 4;1:CD012693. doi: 10.1002/14651858.CD012693.pub3. PMID: 29388198 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
Cited by
-
Mitochondrial Quality Control in Bovine Oocyte Maturation: Mechanisms, Challenges, and Prospects for Enhancing Reproductive Efficiency.Animals (Basel). 2025 Jul 7;15(13):2000. doi: 10.3390/ani15132000. Animals (Basel). 2025. PMID: 40646899 Free PMC article. Review.
-
An overview on cardiac regeneration revolution: exploring the promise of stem cell therapies.Mol Biol Rep. 2025 May 28;52(1):511. doi: 10.1007/s11033-025-10580-6. Mol Biol Rep. 2025. PMID: 40434692 Review.
References
-
- Belva F, Blockeel C, Keymolen K, Buysse A, Bonduelle M, Verheyen G, Roelants M, Tournaye H, Hes F, Van Landuyt L. Impact of embryo vitrification on children’s health, including growth up to two years of age, in comparison with results following a fresh embryo transfer. Fertil Steril 2023;119:932–941. - PubMed
-
- Belva F, Bonduelle M, Roelants M, Michielsen D, Van Steirteghem A, Verheyen G, Tournaye H. Semen quality of young adult ICSI offspring: the first results. Hum Reprod 2016;31:2811–2820. - PubMed
LinkOut - more resources
Full Text Sources
Medical
Research Materials
