Exocyst complex component 1 (Exoc1) loss in dormant oocyte disrupts c-KIT and growth differentiation factor (GDF9) subcellular localization and causes female infertility in mice
- PMID: 39833146
- PMCID: PMC11747099
- DOI: 10.1038/s41420-025-02291-5
Exocyst complex component 1 (Exoc1) loss in dormant oocyte disrupts c-KIT and growth differentiation factor (GDF9) subcellular localization and causes female infertility in mice
Abstract
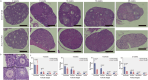
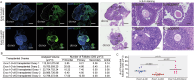
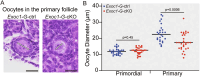
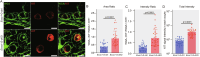
A limited number of female germ cells support reproduction in many mammals. The follicle, composed of oocytes and supporting granulosa cells, forms the basis of oogenesis. Crosstalk between oocytes and granulosa cells is essential for the formation, dormancy, re-awakening, and maturation of oocytes. The oocyte expresses c-KIT and growth differentiation factor-9 (GDF-9), which are major factors in this crosstalk. The downstream signalling pathways of c-KIT and GDF-9 have been well-documented; however, their intra-oocyte trafficking pathway remains unclear. Our study reveals that the exocyst complex, a heterotetrameric protein complex important for tethering in vesicular transport, is important for proper intra-oocyte trafficking of c-KIT and GDF9 in mice. We found that depletion of oocyte-specific EXOC1, a component of the exocyst complex, impaired oocyte re-awakening and cyst breakdown, and inhibited granulosa cell proliferation during follicle growth. The c-KIT receptor is localised on the oocyte plasma membrane. The oocyte-specific Kit conditional knockout mice were reported to exhibit impaired oocyte re-awakening and reduced oocyte cyst breakdown. GDF9 is a protein secreted extracellularly in the oocyte. Previous studies have shown that Gdf9 knockout mice impaired proliferation and granulosa cell multilayering in growing follicles. We found that both c-KIT and GDF9 abnormally stuck in the EXOC1-depleted oocyte cytoplasm. These abnormal phenotypes were also observed in oocytes depleted of exocyst complex members EXOC3 and EXOC7. These results clearly show that the exocyst complex is essential for proper intra-oocyte trafficking of c-KIT and GDF9. Inhibition of this complex causes complete loss of female fertility in mice. Our findings build a platform for research related to trafficking mechanisms of vital crosstalk factors for oogenesis.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics: Animal experimentation: All animal experiments were conducted in a humane manner with approval from the Institutional Animal Experiment Committee of the University of Tsukuba according to the Regulations for Animal Experiments of the University of Tsukuba and the Fundamental Guidelines for Proper Conduct of Animal Experiments and Related Activities in Academic Research Institutions under the jurisdiction of the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Figures

Similar articles
-
Deletion of Exoc7, but not Exoc3, in male germ cells causes severe spermatogenesis failure with spermatocyte aggregation in mice.Exp Anim. 2024 Jul 9;73(3):286-292. doi: 10.1538/expanim.23-0171. Epub 2024 Feb 7. Exp Anim. 2024. PMID: 38325858 Free PMC article.
-
KIT in oocytes: a key factor for oocyte survival and reproductive lifespan.EBioMedicine. 2024 Aug;106:105263. doi: 10.1016/j.ebiom.2024.105263. Epub 2024 Jul 26. EBioMedicine. 2024. PMID: 39067135 Free PMC article.
-
Growth and differentiation factor 9 promotes oocyte growth at the primary but not the early secondary stage in three-dimensional follicle culture.J Assist Reprod Genet. 2016 Aug;33(8):1067-77. doi: 10.1007/s10815-016-0719-z. Epub 2016 May 7. J Assist Reprod Genet. 2016. PMID: 27155601 Free PMC article.
-
Regulation of cumulus expansion and hyaluronan synthesis in porcine oocyte-cumulus complexes during in vitro maturation.Endocr Regul. 2012 Oct;46(4):225-35. doi: 10.4149/endo_2012_04_225. Endocr Regul. 2012. PMID: 23127506 Review.
-
Interaction between growing oocytes and granulosa cells in vitro.Reprod Med Biol. 2019 Aug 22;19(1):13-23. doi: 10.1002/rmb2.12292. eCollection 2020 Jan. Reprod Med Biol. 2019. PMID: 31956281 Free PMC article. Review.
Cited by
-
Current Status of In Vitro Oocyte Growth and Development in Mammals.Reprod Med Biol. 2025 Jul 17;24(1):e12669. doi: 10.1002/rmb2.12669. eCollection 2025 Jan-Dec. Reprod Med Biol. 2025. PMID: 40678565 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

