Engineered CRISPR-Cas9 for Streptomyces sp. genome editing to improve specialized metabolite production
- PMID: 39833194
- PMCID: PMC11747077
- DOI: 10.1038/s41467-025-56278-y
Engineered CRISPR-Cas9 for Streptomyces sp. genome editing to improve specialized metabolite production
Abstract
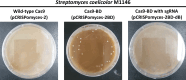
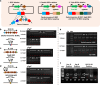
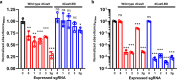
The CRISPR-Cas9 system has frequently been used for genome editing in Streptomyces; however, cytotoxicity, caused by off-target cleavage, limits its application. In this study, we implement innovative modification to Cas9, strategically addressing challenges encountered during gene manipulation using Cas9 within strains possessing high GC content genome. The Cas9-BD, a modified Cas9 with the addition of polyaspartate to its N- and C-termini, is developed with decreased off-target binding and cytotoxicity compared with wild-type Cas9. Cas9-BD and similarly modified dCas9-BD have been successfully employed for simultaneous biosynthetic gene cluster (BGC) refactoring, multiple BGC deletions, or multiplexed gene expression modulations in Streptomyces. We also demonstrate improved secondary metabolite production using multiplexed genome editing with multiple single guide RNA libraries in several Streptomyces strains. Cas9-BD is also used to capture large BGCs using a developed in vivo cloning method. The modified CRISPR-Cas9 system is successfully applied to many Streptomyces sp., providing versatile and efficient genome editing tools for strain engineering of actinomycetes with high GC content genome.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures




Similar articles
-
Application of Cas12j for Streptomyces Editing.Biomolecules. 2024 Apr 16;14(4):486. doi: 10.3390/biom14040486. Biomolecules. 2024. PMID: 38672502 Free PMC article.
-
Fine-Tuning Cas9 Activity with a Cognate Inhibitor AcrIIA4 to Improve Genome Editing in Streptomyces.ACS Synth Biol. 2021 Nov 19;10(11):2833-2841. doi: 10.1021/acssynbio.1c00141. Epub 2021 Nov 4. ACS Synth Biol. 2021. PMID: 34734710
-
Development of a CRISPR/Cas9D10A Nickase (nCas9)-Mediated Genome Editing Tool in Streptomyces.ACS Synth Biol. 2023 Oct 20;12(10):3114-3123. doi: 10.1021/acssynbio.3c00466. Epub 2023 Sep 18. ACS Synth Biol. 2023. PMID: 37722085
-
Genome Editing with CRISPR-Cas9: Can It Get Any Better?J Genet Genomics. 2016 May 20;43(5):239-50. doi: 10.1016/j.jgg.2016.04.008. Epub 2016 Apr 24. J Genet Genomics. 2016. PMID: 27210042 Free PMC article. Review.
-
Challenges and Advances in Genome Editing Technologies in Streptomyces.Biomolecules. 2020 May 8;10(5):734. doi: 10.3390/biom10050734. Biomolecules. 2020. PMID: 32397082 Free PMC article. Review.
Cited by
-
Engineering Useful Microbial Species for Pharmaceutical Applications.Microorganisms. 2025 Mar 5;13(3):599. doi: 10.3390/microorganisms13030599. Microorganisms. 2025. PMID: 40142492 Free PMC article. Review.
-
Exploring pigment-producing Streptomyces as an alternative source to synthetic pigments: diversity, biosynthesis, and biotechnological applications. A review.World J Microbiol Biotechnol. 2025 Jun 25;41(7):211. doi: 10.1007/s11274-025-04379-7. World J Microbiol Biotechnol. 2025. PMID: 40555885 Review.
-
Targeting Siderophore Biosynthesis to Thwart Microbial Growth.Int J Mol Sci. 2025 Apr 11;26(8):3611. doi: 10.3390/ijms26083611. Int J Mol Sci. 2025. PMID: 40332123 Free PMC article. Review.
References
-
- Takahashi, K., Yoshihara, T. & Kurosawa, K. Ushikulides A and B, immunosuppressants produced by a strain of Streptomyces sp. J. Antibiot. (Tokyo)58, 420–424 (2005). - PubMed
-
- Noomnual, S., Thasana, N., Sungkeeree, P., Mongkolsuk, S. & Loprasert, S. Streptanoate, a new anticancer butanoate from Streptomyces sp. DC3. J. Antibiot. (Tokyo)69, 124–127 (2016). - PubMed
-
- Matselyukh, B. et al. N-methylphenylalanyl-dehydrobutyrine diketopiperazine, an A-factor mimic that restores antibiotic biosynthesis and morphogenesis in Streptomyces globisporus 1912-B2 and Streptomyces griseus 1439. J. Antibiot. (Tokyo)68, 9–14 (2015). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

