Impact of protein intake from a caloric-restricted diet on liver lipid metabolism in overweight and obese rats of different sexes
- PMID: 39833384
- PMCID: PMC11747403
- DOI: 10.1038/s41598-025-86596-6
Impact of protein intake from a caloric-restricted diet on liver lipid metabolism in overweight and obese rats of different sexes
Abstract
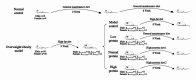
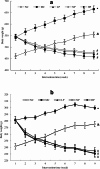
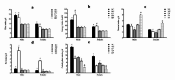
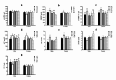
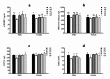
In addition to being linked to an excess of lipid accumulation in the liver, being overweight or obese can also result in disorders of lipid metabolism. There is limited understanding regarding whether different levels of protein intake within an energy-restricted diet affect liver lipid metabolism in overweight and obese rats and whether these effects differ by gender, despite the fact that both high protein intake and calorie restriction can improve intrahepatic lipid. The purpose of this study is to explore the effects and mechanisms of different protein intakes within a calorie-restricted diet on liver lipid metabolism, and to investigate whether these effects exhibit gender differences. The Sprague-Dawley rats, which were half female and half male, were used to construct a rat model of overweight and obesity attributed to a high-fat diet. They were then split up into five groups: the normal control (NC) group, the model control (MC) group, the calorie-restricted low protein (LP) group, the calorie-restricted normal protein (NP) group, and the calorie-restricted high protein (HP) group. Body weight was measured weekly. Samples of plasma and liver were obtained after eight weeks and analyzed for glucose, triglycerides, cholesterol, and hormones in the plasma as well as the liver fat and factors involved in the liver's synthesis and degradation. For the male rats, compared to the HP group, the weight of liver fat in the LP and NP group was significantly higher (P < 0.05). However, for the female rats, there was no significant variation among the three calorie-restricted groups (P > 0.05). There was no significant variation in the concentration of total cholesterol (TC), very low density lipoprotein (VLDL), low density lipoprotein (LDL), and high density lipoprotein (HDL) among the three male calorie-restricted groups (P > 0.05), while the TC and VLDL concentrations in the female LP and NP group were significantly higher compared to those in the HP group (P < 0.05). Moreover, the trend of expression in the signaling pathways of adiponectin/phosphorylated AMP-activated protein kinase (p-AMPK) and adiponectin/peroxisome proliferators-activated receptor alpha (PPARα) in the liver was consistent with that of the liver fat content, and leptin acted in the same way as adiponectin. Compared with the three calorie-restricted groups, the expressions of nuclear sterol-regulatory element-binding protein-2 (nSREBP-2) and 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG-CoA reductase) involved in cholesterol synthesis and low-density lipoprotein receptor (LDLR) and cholesterol 7-alpha hydroxylase (CYP7A1) involved in cholesterol clearance in the MC group were significantly lower (P < 0.05). A 40% energy restriction can significantly reduce the body weight, body fat, liver fat, and the blood concentration of TG in both male and female overweight and obese rats, but it can significantly increase the blood concentration of TC in overweight and obese male rats. At the same time of 40% calorie restriction, increasing dietary protein intake to twice the normal protein intake has a stronger effect on promoting hepatic triglyceride oxidation and reducing liver fat content in the male overweight and obese rats by increasing the levels of adiponectin and leptin in the blood, and can also significantly reduce the plasma cholesterol concentration in the female overweight and obese rats through inhibiting cholesterol synthesis most likely by increasing glucagon level in the blood.
Keywords: Calorie restriction; Lipid metabolism; Liver; Obesity; Overweight; Protein intake; Rat.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures
Similar articles
-
Effects of protein intake from an energy-restricted diet on the skeletal muscle composition of overweight and obese rats.Sci Rep. 2022 Nov 27;12(1):20396. doi: 10.1038/s41598-022-24961-5. Sci Rep. 2022. PMID: 36437293 Free PMC article.
-
Methionine restriction prevents the progression of hepatic steatosis in leptin-deficient obese mice.Metabolism. 2013 Nov;62(11):1651-61. doi: 10.1016/j.metabol.2013.06.012. Epub 2013 Aug 5. Metabolism. 2013. PMID: 23928105
-
Ethanol Extract of Peanut Sprout Lowers Blood Triglyceride Levels, Possibly Through a Pathway Involving SREBP-1c in Rats Fed a High-Fat Diet.J Med Food. 2015 Aug;18(8):850-5. doi: 10.1089/jmf.2014.3370. Epub 2015 May 6. J Med Food. 2015. PMID: 25946626
-
Leucine improves protein nutritional status and regulates hepatic lipid metabolism in calorie-restricted rats.Cell Biochem Funct. 2014 Jun;32(4):326-32. doi: 10.1002/cbf.3017. Epub 2013 Dec 11. Cell Biochem Funct. 2014. PMID: 24327520
-
Unlocking the potential: How acupuncture reshapes the liver-centered lipid metabolism pattern to fight obesity.J Integr Med. 2024 Sep;22(5):523-532. doi: 10.1016/j.joim.2024.08.004. Epub 2024 Aug 20. J Integr Med. 2024. PMID: 39209583 Review.
References
-
- Drummen, M. et al. Long-term effects of increased protein intake after weight loss on intrahepatic lipid content and implications for insulin sensitivity: a PREVIEW study. Am. J. Physiol. Endocrinol. Metab.315, 885–891. 10.1152/ajpendo.00162.2018 (2018). - PubMed
-
- Yu, H., Nie, R. & Shen, C. The role of bile acids in regulating glucose and lipid metabolism. Endocr. J.70, 359–374. 10.1507/endocrj.EJ22-0544 (2023). - PubMed
-
- Nagarajan, S. R., Cross, E., Sanna, F. & Hodson, L. Dysregulation of hepatic metabolism with obesity: factors influencing glucose and lipid metabolism. Proc. Nutr. Soc.81, 1–11. 10.1017/s0029665121003761 (2022). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

