Muscular TOR knockdown and endurance exercise ameliorate high salt and age-related skeletal muscle degradation by activating the MTOR-mediated pathway
- PMID: 39841657
- PMCID: PMC11753686
- DOI: 10.1371/journal.pone.0311159
Muscular TOR knockdown and endurance exercise ameliorate high salt and age-related skeletal muscle degradation by activating the MTOR-mediated pathway
Abstract
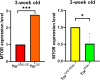
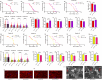
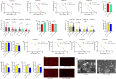
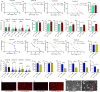
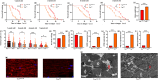
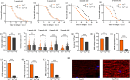
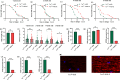
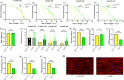
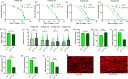
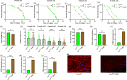
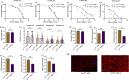
The target of rapamycin(TOR)gene is closely related to metabolism and cellular aging, but it is unclear whether the TOR pathways mediate endurance exercise against the accelerated aging of skeletal muscle induced by high salt intake. In this study, muscular TOR gene overexpression and RNAi were constructed by constructing MhcGAL4/TOR-overexpression and MhcGAL4/TORUAS-RNAi systems in Drosophila. The results showed that muscle TOR knockdown and endurance exercise significantly increased the climbing speed, climbing endurance, the expression of autophagy related gene 2(ATG2), silent information regulator 2(SIR2), and pparγ coactivator 1(PGC-1α) genes, and superoxide dismutases(SOD) activity, but it decreased the expression of the TOR gene and reactive oxygen species(ROS) level, and it protected the myofibrillar fibers and mitochondria of skeletal muscle in Drosophila on a high-salt diet. TOR overexpression yielded similar results to the high salt diet(HSD) alone, with the opposite effect of TOR knockout found in regard to endurance exercise and HSD-induced age-related skeletal muscle degradation. Therefore, the current findings confirm that the muscle TOR gene plays an important role in endurance exercise against HSD-induced age-related skeletal muscle degeneration, as it determines the activity of the mammalian target of rapamycin(MTOR)/SIR2/PGC-1α and MTOR/ATG2/PGC-1α pathways in skeletal muscle.
Copyright: © 2025 Wang et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Muscle-specific overexpression of Atg2 gene and endurance exercise delay age-related deteriorations of skeletal muscle and heart function via activating the AMPK/Sirt1/PGC-1α pathway in male Drosophila.FASEB J. 2023 Nov;37(11):e23214. doi: 10.1096/fj.202301312R. FASEB J. 2023. PMID: 37773768
-
Physical exercise ameliorates age-related deterioration of skeletal muscle and mortality by activating Pten-related pathways in Drosophila on a high-salt diet.FASEB J. 2023 Dec;37(12):e23304. doi: 10.1096/fj.202301099R. FASEB J. 2023. PMID: 37971426
-
Endurance exercise attenuates Gαq-RNAi induced hereditary obesity and skeletal muscle dysfunction via improving skeletal muscle Srl/MRCC-I pathway in Drosophila.Sci Rep. 2024 Nov 15;14(1):28207. doi: 10.1038/s41598-024-79415-x. Sci Rep. 2024. PMID: 39548180 Free PMC article.
-
Integrative effects of resistance training and endurance training on mitochondrial remodeling in skeletal muscle.Eur J Appl Physiol. 2024 Oct;124(10):2851-2865. doi: 10.1007/s00421-024-05549-5. Epub 2024 Jul 9. Eur J Appl Physiol. 2024. PMID: 38981937 Review.
-
Autophagy is essential to support skeletal muscle plasticity in response to endurance exercise.Am J Physiol Regul Integr Comp Physiol. 2014 Oct 15;307(8):R956-69. doi: 10.1152/ajpregu.00187.2014. Epub 2014 Aug 13. Am J Physiol Regul Integr Comp Physiol. 2014. PMID: 25121614 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

