S100A8-CAMKK2-AMPK axis confers the protective effects of mild hypothermia against cerebral ischemia-reperfusion injury in rats
- PMID: 39843475
- PMCID: PMC11754893
- DOI: 10.1038/s41598-025-87184-4
S100A8-CAMKK2-AMPK axis confers the protective effects of mild hypothermia against cerebral ischemia-reperfusion injury in rats
Abstract
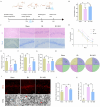
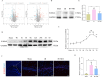
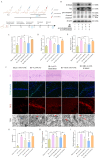
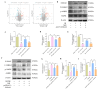
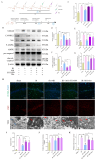
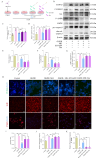
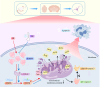
To investigate the neuroprotective mechanism of mild hypothermia (MH) in ameliorating cerebral ischemia reperfusion (IR) injury. The Pulsinelli's four-vessel ligation method was utilized to establish a rat model of global cerebral IR injury. To investigate the role of S100A8 in MH treatment of cerebral IR injury, hippocampus-specific S100A8 loss or gain of function was achieved using an adeno-associated virus system. We examined the effect of S100A8 over-expression or knock-down on the function of the SH-SY5Y cell line subjected to oxygen-glucose deprivation reoxygenation (OGDR) injury under MH treatment and delved into the underlying mechanisms. MH significantly ameliorates IR-induced neurological injury in the brain. Similarly to MH, knock-down of S100A8 significantly reduced neuronal oxidative stress, attenuated mitochondrial damage, inhibited apoptosis, and improved cognitive function in IR rats. Conversely, over-expression of S100A8 attenuated MH's protective effect and aggravated brain IR injury. In vitro, low expression of S100A8 significantly inhibited the decline in mitochondrial membrane potential induced by OGDR, reduced oxidative stress response, and decreased cell apoptosis, acting as a protective agent nearly equivalent to MH in SH-SY5Y cells. However, over-expression of S100A8 significantly inhibited these protective effects of MH. Mechanistically, MH down-regulated S100A8 expression, enhancing mitochondrial function via activation of the CAMKK2/AMPK signaling pathway. Moreover, with MH treatment, the administration of CAMKK2 and AMPK inhibitors STO-609 and Dorsomorphin significantly increased oxidative stress, mitochondrial damage, and cell apoptosis, thereby diminishing MH's neuroprotective effect against cerebral IR injury. Our study identified S100A8 as a master regulator that enables MH to ameliorate neurological injury during the early stage of cerebral IR injury by enhancing mitochondrial function. By targeting the S100A8-initiated CAMKK2/AMPK signaling pathway, we may unlock a novel therapeutic intervention or develop a refined MH therapeutic strategy against cerebral IR injury.
Keywords: Apoptosis; CAMKK2; Cerebral ischemia reperfusion; Mild hypothermia; S100A8.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: Male Wistar Rats (body weight: 250–300 g, age: 6–8 weeks) were obtained from Jinan Pengyue Laboratory Animal Breeding Company (Jinan, China). The animal experiments were reviewed and approved by the Qingdao University Medical Ethics Committee (QDU-AEC-2024473). Consent for publication: Not applicable. Statement on ARRIVE guidelines: We declared that this study was carried out in compliance with the ARRIVE guidelines.
Figures

Similar articles
-
miR-378a-5p regulates CAMKK2/AMPK pathway to contribute to cerebral ischemia/reperfusion injury-induced neuronal apoptosis.Folia Histochem Cytobiol. 2021;59(1):57-65. doi: 10.5603/FHC.a2021.0007. Epub 2021 Mar 2. Folia Histochem Cytobiol. 2021. PMID: 33651374
-
Mild Hypothermia Combined with Hydrogen Sulfide Treatment During Resuscitation Reduces Hippocampal Neuron Apoptosis Via NR2A, NR2B, and PI3K-Akt Signaling in a Rat Model of Cerebral Ischemia-Reperfusion Injury.Mol Neurobiol. 2016 Sep;53(7):4865-73. doi: 10.1007/s12035-015-9391-z. Epub 2015 Sep 9. Mol Neurobiol. 2016. PMID: 26350917
-
Proteome profiling of hippocampus reveals the neuroprotective effect of mild hypothermia on global cerebral ischemia-reperfusion injury in rats.Sci Rep. 2023 Sep 2;13(1):14450. doi: 10.1038/s41598-023-41766-2. Sci Rep. 2023. PMID: 37660166 Free PMC article.
-
Inhibition of autophagy by CRMP2-derived peptide ST2-104 (R9-CBD3) via a CaMKKβ/AMPK/mTOR pathway contributes to ischemic postconditioning-induced neuroprotection against cerebral ischemia-reperfusion injury.Mol Brain. 2021 Aug 6;14(1):123. doi: 10.1186/s13041-021-00836-0. Mol Brain. 2021. PMID: 34362425 Free PMC article.
-
Factors Contributing to Resistance to Ischemia-Reperfusion Injury in Olfactory Mitral Cells.Int J Mol Sci. 2025 May 25;26(11):5079. doi: 10.3390/ijms26115079. Int J Mol Sci. 2025. PMID: 40507890 Free PMC article. Review.
References
-
- Fanning, J. P. et al. Perioperative stroke. Nat. Rev. Dis. Primers10, 3. 10.1038/s41572-023-00487-6 (2024). - PubMed
-
- Li, W. et al. Near-infrared-II imaging revealed hypothermia regulates neuroinflammation following brain injury by increasing the glymphatic influx. ACS Nano18, 13836–13848. 10.1021/acsnano.4c02652 (2024). - PubMed
-
- Polderman, K. H. Mechanisms of action, physiological effects, and complications of hypothermia. Crit. Care Med.37, S186-202. 10.1097/CCM.0b013e3181aa5241 (2009). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

