Multimodal treatments and the risk of breast cancer-related lymphedema: insights from a nationally representative cohort in South Korea
- PMID: 39844110
- PMCID: PMC11753110
- DOI: 10.1186/s12885-025-13513-5
Multimodal treatments and the risk of breast cancer-related lymphedema: insights from a nationally representative cohort in South Korea
Abstract
Background: As the roles of chemotherapy (Chemo) and radiation therapy (Radio) in the definitive treatment of breast cancer have expanded, a broader understanding of the factors associated with breast cancer-related lymphedema (BCRL) has become increasingly essential. Therefore, we investigated the association between multimodality treatment and the risk of BCRL.
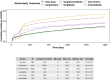
Methods: In this retrospective study conducted using National Health Insurance data and the Korea National Cancer Incidence Database (2006-2017), 114,638 participants who underwent Surgery (Surg) or Chemo within 6 months after breast cancer diagnosis were enrolled, and the effect of multimodality treatment on the risk of BCRL was analyzed using the Cox proportional-hazards model. Multimodality treatment administered through six months of treatment was grouped as only Surg; Surg/Chemo; Surg/ Chemo/Radio; Surg/Radio; only Chemo; and Chemo/Radio.
Results: The risk of BCRL was higher in the Surg/Chemo group (hazard ratio [HR]: 1.54, 95% confidence interval [CI]: 1.43-1.65), Surg/Chemo/Radio group (HR: 1.51, 95% CI: 1.43-1.65), only Chemo group (HR: 1.58, 95% CI: 1.45-1.71), and Chemo/Radio group (HR: 1.13, 95% CI: 1.00-1.29) in comparison with the only Surg group.
Conclusion: BCRL occurrence is an after-effect of complex breast cancer treatments, and the risk may vary depending on the treatment method, including Surg, chemo, and radio. Our findings suggest that multidisciplinary strategies are required to minimize the risk of BCRL development in patients with breast cancer.
Keywords: Breast cancer; Epidemiology; Multimodality treatment; Risk of breast cancer-related lymphedema.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All procedures contributing to this work complied with the ethical standards of the relevant national and institutional committees on human experimentation and the Helsinki Declaration of 1975, as revised in 2008. As the NHIS data did not contain any identifying information, the study was approved by the Institutional Review Board of Seoul National University Hospital (E-2206–076-1332). Because we used de-identified data, the need for informed consent was waived by the Institutional Review Board of Seoul National University Hospital (E-2206–076-1332). Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials