Butyrolactone I blocks the transition of acute kidney injury to chronic kidney disease in mice by targeting JAK1
- PMID: 39845897
- PMCID: PMC11751251
- DOI: 10.1002/mco2.70064
Butyrolactone I blocks the transition of acute kidney injury to chronic kidney disease in mice by targeting JAK1
Erratum in
-
Correction to "Butyrolactone I Blocks the Transition of Acute Kidney Injury to Chronic Kidney Disease in Mice by Targeting JAK1".MedComm (2020). 2025 Dec 9;6(12):e70552. doi: 10.1002/mco2.70552. eCollection 2025 Dec. MedComm (2020). 2025. PMID: 41383496 Free PMC article.
Abstract
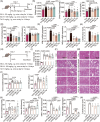
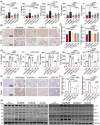
Chronic kidney disease (CKD) is a disease that affects more than 850 million people. Acute kidney injury (AKI) is a common cause of CKD, and blocking the AKI-CKD transition shows promising therapeutic potential. Herein, we found that butyrolactone I (BLI), a natural product, exerts significant nephroprotective effects, including maintenance of kidney function, inhibition of inflammatory response, and prevention of fibrosis, in both folic acid- and ureteral obstruction-induced AKI-CKD transition mouse models. Notably, BLI showed greater blood urea nitrogen reduction and anti-inflammatory effects than telmisartan. Bioinformatics analysis and target confirmation assays suggested that BLI directly binds to JAK1, and kinase inhibition assay confirmed it is a potent JAK1inhibitor with an IC50 of 0.376 µM. Experiments in JAK1-knockdown mice also proved that BLI targets JAK1 to work. Furthermore, BLI demonstrated nephroprotective effects and safety comparable to ivarmacitinib, the well-known JAK1 inhibitor. Mechanistically, BLI targets JAK1 and inhibits its phosphorylation and JAK-STAT activation, subsequently regulating the downstream signaling pathways to inhibit reactive oxygen species production, inflammation, and ferroptosis, thereby preventing the occurrence of kidney fibrosis and blocking the AKI-CKD transition process. This study demonstrates for the first time that BLI is a JAK1 inhibitor and a promising candidate for delaying CKD progression, which warrants further investigation.
Keywords: AKI–CKD transition; JAK1; butyrolactone I; ferroptosis.
© 2025 The Author(s). MedComm published by Sichuan International Medical Exchange & Promotion Association (SCIMEA) and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures






References
-
- Kalantar‐Zadeh K, Jafar TH, Nitsch D, Neuen BL, Perkovic V. Chronic kidney disease. Lancet. 2021;398(10302):786‐802. - PubMed
-
- Kellum JA, Romagnani P, Ashuntantang G, Ronco C, Zarbock A, Anders H‐J. Acute kidney injury. Nat Rev Dis Primers. 2021;7(1):52. - PubMed
-
- Levin A, Ahmed SB, Carrero JJ, et al. Executive summary of the KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease: known knowns and known unknowns. Kidney Int. 2024;105(4):684‐701. - PubMed
-
- Lv J‐C, Zhang L‐X. Prevalence and disease burden of chronic kidney disease. In: Liu B‐C, Lan H‐Y, Lv L‐L, eds. Renal Fibrosis: Mechanisms and Therapies. Springer; 2019:3‐15. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
