Prognostic value of immunosuppression scores in patients with esophageal squamous cell carcinoma: a multicenter study
- PMID: 39845968
- PMCID: PMC11752912
- DOI: 10.3389/fimmu.2024.1517968
Prognostic value of immunosuppression scores in patients with esophageal squamous cell carcinoma: a multicenter study
Abstract
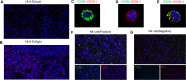
Introduction: The prognostic impact of human leukocyte antigen-E (HLA-E) expression and the proportion of natural killer (NK) cells in esophageal squamous cell carcinoma (ESCC) was investigated.
Methods: This study retrospectively evaluated 397 ESCC patients across two centers. The cumulative incidence of recurrence (CIR) and the incidence of tumor-related death (CID) were analyzed in various groups. An immunosuppression score (ISS) was developed based on HLA-E expression and NK cell proportion. Differences between groups were adjusted using inverse probability treatment weighting (IPTW). The factors influencing cancer-specific survival (CSS) and recurrence-free survival (RFS) were also examined.
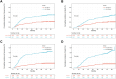
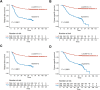
Results: Patients with low HLA-E expression had significantly higher five-year CIR and CID compared to those with high expression (CIR: 20.7% vs. 45.1%, CID: 19.3% vs. 40.1%; p< 0.001). Similarly, NK cell-positive patients had significantly better five-year CIR and CID than NK cell-negative patients (CIR: 16.3% vs. 59.6%, CID: 13.9% vs. 53.7%; p < 0.001). The Sankey diagram indicated that the low ISS group had a lower recurrence and tumor-related mortality rate (p < 0.05). After IPTW adjustment, the low ISS group showed improved five-year RFS (80.1% vs. 35.4%, p < 0.001) and five-year CSS (82.3% vs. 42.5%, p < 0.001) compared to the high ISS group.
Conclusions: ESCC with different ISS statuses represents two distinct biological subtypes, underscoring the need for personalized treatment strategies tailored to varying tumor behaviors.
Keywords: esophageal squamous cell carcinoma (ESCC); human leukocyte antigen-E (HLA-E); inverse probability treatment weighting (IPTW); natural killer (NK) cells; prognosis.
Copyright © 2025 Xu, Luo, Huang, Tu, Chen, Shen, Sun and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Xu J, Kato K, Raymond E, Hubner RA, Shu Y, Pan Y, et al. Tislelizumab plus chemotherapy versus placebo plus chemotherapy as first-line treatment for advanced or metastatic oesophageal squamous cell carcinoma (RATIONALE-306): a global, randomised, placebo-controlled, phase 3 study. Lancet Oncol. (2023) 24(5):483–95. doi: 10.1016/S1470-2045(23)00108-0 - DOI - PubMed
-
- Sun JM, Shen L, Shah MA, Enzinger P, Adenis A, Doi T, et al. Pembrolizumab plus chemotherapy versus chemotherapy alone for first-line treatment of advanced oesophageal cancer (KEYNOTE-590): a randomised, placebo-controlled, phase 3 study. Lancet. (2021) 398(10302):759–71. doi: 10.1016/S0140-6736(21)01234-4 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

