Investigation of the threonine metabolism of Echinococcus multilocularis: The threonine dehydrogenase as a potential drug target in alveolar echinococcosis
- PMID: 39847910
- PMCID: PMC11795093
- DOI: 10.1016/j.ijpddr.2025.100581
Investigation of the threonine metabolism of Echinococcus multilocularis: The threonine dehydrogenase as a potential drug target in alveolar echinococcosis
Abstract
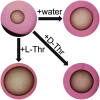
Alveolar echinococcosis (AE) is a severe zoonotic disease caused by the metacestode stage of the fox tapeworm Echinococcus multilocularis. We recently showed that E. multilocularis metacestode vesicles scavenge large amounts of L-threonine from the culture medium. This motivated us to study the effect of L-threonine on the parasite and how it is metabolized. We established a novel metacestode vesicle growth assay with an automated readout, which showed that L-threonine treatment led to significantly increased parasite growth. In addition, L-threonine increased the formation of novel metacestode vesicles from primary parasite cell cultures in contrast to the non-proteinogenic threonine analog 3-hydroxynorvaline. Tracing of [U-13C]-L-threonine and metabolites in metacestode vesicles and culture medium resulted in the detection of [U-13C]-labeling in aminoacetone and glycine, indicating that L-threonine was metabolized by threonine dehydrogenase (TDH). EmTDH-mediated threonine metabolism in the E. multilocularis metacestode stage was further confirmed by quantitative real-time PCR, which demonstrated high expression of emtdh in in vitro cultured metacestode vesicles and also in metacestode samples obtained from infected animals. EmTDH was enzymatically active in metacestode vesicle extracts. The compounds disulfiram, myricetin, quercetin, sanguinarine, and seven quinazoline carboxamides were evaluated for their ability to inhibit recombinantly expressed EmTDH. The most potent inhibitors, albeit not very strong or highly specific, were disulfiram, myricetin and sanguinarine. These compounds were subsequently tested for activity against E. multilocularis metacestode vesicles and primary parasite cells and only sanguinarine demonstrated significant in vitro activity. However, TDH is not its only cellular target, and it is also known to be highly toxic. Our findings suggest that additional targets of sanguinarine should be explored, and that it may serve as a foundation for developing more specific compounds against the parasite. Moreover, the EmTDH assay could be a valuable high-throughput, target-based platform for discovering novel anti-echinococcal compounds.
Keywords: Cestode; Disulfiram; Echinococcus multilocularis; Sanguinarine; Target-based screening; Threonine metabolism.
Copyright © 2025 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures








Similar articles
-
EmTIP, a T-Cell immunomodulatory protein secreted by the tapeworm Echinococcus multilocularis is important for early metacestode development.PLoS Negl Trop Dis. 2014 Jan 2;8(1):e2632. doi: 10.1371/journal.pntd.0002632. eCollection 2014. PLoS Negl Trop Dis. 2014. PMID: 24392176 Free PMC article.
-
Host insulin stimulates Echinococcus multilocularis insulin signalling pathways and larval development.BMC Biol. 2014 Jan 27;12:5. doi: 10.1186/1741-7007-12-5. BMC Biol. 2014. PMID: 24468049 Free PMC article.
-
A MEKK1 - JNK mitogen activated kinase (MAPK) cascade module is active in Echinococcus multilocularis stem cells.PLoS Negl Trop Dis. 2021 Dec 8;15(12):e0010027. doi: 10.1371/journal.pntd.0010027. eCollection 2021 Dec. PLoS Negl Trop Dis. 2021. PMID: 34879059 Free PMC article.
-
Molecular survival strategies of Echinococcus multilocularis in the murine host.Parasitol Int. 2006;55 Suppl:S45-9. doi: 10.1016/j.parint.2005.11.006. Epub 2005 Dec 13. Parasitol Int. 2006. PMID: 16352460 Review.
-
Treatment of echinococcosis: albendazole and mebendazole--what else?Parasite. 2014;21:70. doi: 10.1051/parasite/2014073. Epub 2014 Dec 22. Parasite. 2014. PMID: 25526545 Free PMC article. Review.
Cited by
-
Host Proteins in Echinococcus multilocularis Metacestodes.Int J Mol Sci. 2025 Apr 1;26(7):3266. doi: 10.3390/ijms26073266. Int J Mol Sci. 2025. PMID: 40244114 Free PMC article.
References
-
- Adjogatse E., Erskine P., Wells S.A., Kelly J.M., Wilden J.D., Chan A.W.E., Selwood D., Coker A., Wood S., Cooper J.B. Structure and function of L-threonine-3-dehydrogenase from the parasitic protozoan Trypanosoma brucei revealed by X-ray crystallography and geometric simulations. Acta Crystallogr. Sect. Struct. Biol. 2018;74:861–876. doi: 10.1107/S2059798318009208. - DOI - PubMed
-
- Adjogatse E.K. Structure-based drug design for the discovery of new treatments for trypanosomiasis. Dr. Thesis UCL Univ. Coll. Lond. UCL (University College London) 2015 https://discovery.ucl.ac.uk/id/eprint/1467152/
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources

