Pharmacological and structural insights into nanvuranlat, a selective LAT1 (SLC7A5) inhibitor, and its N-acetyl metabolite with implications for cancer therapy
- PMID: 39849059
- PMCID: PMC11758009
- DOI: 10.1038/s41598-025-87522-6
Pharmacological and structural insights into nanvuranlat, a selective LAT1 (SLC7A5) inhibitor, and its N-acetyl metabolite with implications for cancer therapy
Abstract
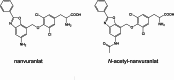
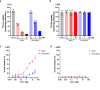
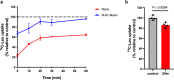
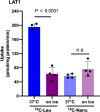
L-type amino acid transporter 1 (LAT1, SLC7A5), overexpressed in various cancers, mediates the uptake of essential amino acids crucial for tumor growth. It has emerged as a promising target for cancer therapy. Nanvuranlat (JPH203/KYT-0353), a LAT1 inhibitor, has shown antitumor activity in preclinical studies and efficacy in biliary tract cancer during clinical trials. This study provides a comprehensive pharmacological characterization of nanvuranlat and its N-acetyl metabolite, including structural insights into their LAT1 interactions. Both compounds demonstrated high selectivity for LAT1 over LAT2 and other amino acid transporters. Nanvuranlat acts as a competitive, non-transportable LAT1 inhibitor (Ki = 38.7 nM), while its N-acetyl metabolite retains selectivity but with reduced affinity (Ki = 1.68 µM). Nanvuranlat exhibited a sustained inhibitory effect on LAT1 even after its removal, indicating the potential for prolonged therapeutic effects. Both compounds showed comparable dissociation rates, suggesting that N-acetylation does not affect the interaction responsible for slow dissociation. The U-shaped conformation adopted by nanvuranlat when bound to LAT1 likely contributes to its high affinity, selectivity, sustained inhibitory effect, and non-transportable nature observed in this study. These insights into nanvuranlat's mechanism and metabolic impact provide essential information for understanding its clinical efficacy and advancing LAT1-targeted cancer therapies.
Keywords: Amino acid transporter; Cancer therapy; Drug metabolism & efficacy; LAT1 inhibitor; Pharmacological selectivity.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Combination effects of amino acid transporter LAT1 inhibitor nanvuranlat and cytotoxic anticancer drug gemcitabine on pancreatic and biliary tract cancer cells.Cancer Cell Int. 2023 Jun 15;23(1):116. doi: 10.1186/s12935-023-02957-z. Cancer Cell Int. 2023. PMID: 37322479 Free PMC article.
-
Structural basis of anticancer drug recognition and amino acid transport by LAT1.Nat Commun. 2025 Feb 14;16(1):1635. doi: 10.1038/s41467-025-56903-w. Nat Commun. 2025. PMID: 39952931 Free PMC article.
-
Amino acid transporter LAT1 (SLC7A5) as a molecular target for cancer diagnosis and therapeutics.Pharmacol Ther. 2022 Feb;230:107964. doi: 10.1016/j.pharmthera.2021.107964. Epub 2021 Aug 12. Pharmacol Ther. 2022. PMID: 34390745 Review.
-
Upregulation of ATF4 mediates the cellular adaptation to pharmacologic inhibition of amino acid transporter LAT1 in pancreatic ductal adenocarcinoma cells.J Pharmacol Sci. 2024 May;155(1):14-20. doi: 10.1016/j.jphs.2024.03.001. Epub 2024 Mar 13. J Pharmacol Sci. 2024. PMID: 38553134
-
The Role of Large Neutral Amino Acid Transporter (LAT1) in Cancer.Curr Cancer Drug Targets. 2019;19(11):863-876. doi: 10.2174/1568009619666190802135714. Curr Cancer Drug Targets. 2019. PMID: 31376820 Review.
References
-
- Christensen, H. N. Role of amino acid transport and countertransport in nutrition and metabolism. Physiol. Rev.70, 43–77 (1990). - PubMed
-
- Fotiadis, D., Kanai, Y. & Palacín, M. The SLC3 and SLC7 families of amino acid transporters. Mol. Aspects Med.34, 139–158 (2013). - PubMed
-
- Kanai, Y. Amino acid transporter LAT1 (SLC7A5) as a molecular target for cancer diagnosis and therapeutics. Pharmacol. Ther.230, 107964 (2022). - PubMed
-
- Kanai, Y. et al. Expression cloning and characterization of a transporter for large neutral amino acids activated by the heavy chain of 4F2 antigen (CD98). J. Biol. Chem.273, 23629–23632 (1998). - PubMed
-
- Mastroberardino, L. et al. Amino-acid transport by heterodimers of 4F2hc/CD98 and members of a permease family. Nature395, 288–291 (1998). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

