Molecular Mechanisms of Nicergoline from Ergot Fungus in Blocking Human 5-HT3A Receptor
- PMID: 39849925
- PMCID: PMC11813358
- DOI: 10.4014/jmb.2411.11020
Molecular Mechanisms of Nicergoline from Ergot Fungus in Blocking Human 5-HT3A Receptor
Abstract
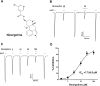
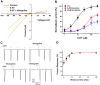
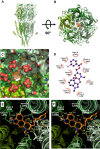
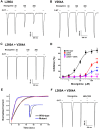
This study investigates the modulatory effects of nicergoline, a major bioactive compound derived from ergot fungus, on the 5-hydroxytryptamine 3A (5-HT3A) receptor. Utilizing a two-electrode voltage-clamp technique, we evaluated the impact of nicergoline on the 5-HT-induced inward current (I5-HT) in 5-HT3A receptors. Our findings reveal that nicergoline inhibits I5-HT in a reversible and concentration-dependent manner. Additionally, the observed voltage-dependent and use-dependent inhibition indicates that nicergoline acts as an open channel blocker of the 5-HT3A receptor. To further elucidate the interaction between nicergoline and the 5-HT3A receptor, we conducted molecular docking studies. Overactivation of the 5-HT3A receptor can enhance excitatory neurotransmission, potentially leading to heightened anxiety and stress responses. It may also interfere with hippocampal functions, adversely affecting learning and memory. Additionally, exceed activation of these receptors is a primary mechanism underlying nausea and vomiting, commonly observed during chemotherapy or in response to certain toxins. Collectively, our results suggest that nicergoline has the potential to inhibit 5-HT3A receptor activity by interacting with binding residues L260 and V264. This inhibition may enhance cognitive function by stabilizing neural circuits involved in cognitive processes and can improve cognitive symptoms in patients with dementia. Additionally, the anxiolytic effects resulting from 5-HT3A receptor inhibition could promote overall psychological well-being in affected individuals. Thus, the role of nicergoline as a 5-HT3A receptor antagonist not only highlights its therapeutic potential but also warrants further exploration into its mechanisms and broader implications for managing neurodegenerative diseases.
Keywords: 5-HT3A receptor; Ergot fungus; nicergoline; open channel blocker.
Conflict of interest statement
The authors have no financial conflicts of interest to declare.
Figures
Similar articles
-
Identification and molecular study on the interaction of Schisandrin C with human 5-HT3A receptor.Eur J Pharmacol. 2021 Sep 5;906:174220. doi: 10.1016/j.ejphar.2021.174220. Epub 2021 May 31. Eur J Pharmacol. 2021. PMID: 34081905
-
Differential Regulation of Human Serotonin Receptor Type 3A by Chanoclavine and Ergonovine.Molecules. 2021 Feb 24;26(5):1211. doi: 10.3390/molecules26051211. Molecules. 2021. PMID: 33668306 Free PMC article.
-
Tripterygium-derived celastrol inhibits 5-HT3A receptors via key residue interactions: A comparative electrophysiological and docking study.Neurosci Lett. 2025 Jul 13;859-861:138265. doi: 10.1016/j.neulet.2025.138265. Epub 2025 May 15. Neurosci Lett. 2025. PMID: 40381881
-
Recent developments in 5-HT3 receptor pharmacology.Trends Pharmacol Sci. 2013 Feb;34(2):100-9. doi: 10.1016/j.tips.2012.12.002. Trends Pharmacol Sci. 2013. PMID: 23380247 Review.
-
The 5-hydroxytryptamine type 3 (5-HT3) receptor reveals a novel determinant of single-channel conductance.Biochem Soc Trans. 2004 Jun;32(Pt3):547-52. doi: 10.1042/BST0320547. Biochem Soc Trans. 2004. PMID: 15157181 Review.
Cited by
-
Phenethyl Acetate as an Agonist of Insect Odorant Receptor Co-Receptor (Orco): Molecular Mechanisms and Functional Insights.Int J Mol Sci. 2025 May 22;26(11):4970. doi: 10.3390/ijms26114970. Int J Mol Sci. 2025. PMID: 40507781 Free PMC article.
References
-
- Bennett J, Bentley R. Pride and prejudice: the story of ergot. Perspect. Biol. Med. 1999;42:333–355. doi: 10.1353/pbm.1999.0026. - DOI
-
- Kren V, Cvak L. Ergot: the genus Claviceps. Ed. CRC Press; 1999. pp. 89–90.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

