Understanding the Immune System and Biospecimen-Based Response in Glioblastoma: A Practical Guide to Utilizing Signal Redundancy for Biomarker and Immune Signature Discovery
- PMID: 39851932
- PMCID: PMC11763554
- DOI: 10.3390/curroncol32010016
Understanding the Immune System and Biospecimen-Based Response in Glioblastoma: A Practical Guide to Utilizing Signal Redundancy for Biomarker and Immune Signature Discovery
Abstract
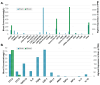
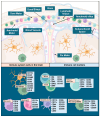
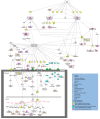
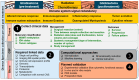
Glioblastoma (GBM) is a primary central nervous system malignancy with a median survival of 15-20 months. The presence of both intra- and intertumoral heterogeneity limits understanding of biological mechanisms leading to tumor resistance, including immune escape. An attractive field of research to examine treatment resistance are immune signatures composed of cluster of differentiation (CD) markers and cytokines. CD markers are surface markers expressed on various cells throughout the body, often associated with immune cells. Cytokines are the effector molecules of the immune system. Together, CD markers and cytokines can serve as useful biomarkers to reflect immune status in patients with GBM. However, there are gaps in the understanding of the intricate interactions between GBM and the peripheral immune system and how these interactions change with standard and immune-modulating treatments. The key to understanding the true nature of these interactions is through multi-omic analysis of tumor progression and treatment response. This review aims to identify potential non-invasive blood-based biomarkers that can contribute to an immune signature through multi-omic approaches, leading to a better understanding of immune involvement in GBM.
Keywords: biomarkers; glioblastoma; immune response; immune system; tumor resistance.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Stupp R., Hegi M.E., Mason W.P., van den Bent M.J., Taphoorn M.J., Janzer R.C., Ludwin S.K., Allgeier A., Fisher B., Belanger K., et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10:459–466. doi: 10.1016/S1470-2045(09)70025-7. - DOI - PubMed
-
- Vaubel R.A., Tian S., Remonde D., Schroeder M.A., Mladek A.C., Kitange G.J., Caron A., Kollmeyer T.M., Grove R., Peng S., et al. Genomic and Phenotypic Characterization of a Broad Panel of Patient-Derived Xenografts Reflects the Diversity of Glioblastoma. Clin. Cancer Res. 2020;26:1094–1104. doi: 10.1158/1078-0432.CCR-19-0909. - DOI - PMC - PubMed
-
- Hanahan D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022;12:31–46. doi: 10.1158/2159-8290.CD-21-1059. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

